Vernakalant blocks both sodium (Na⁺) and potassium (K⁺) channels,
- it does not fit into the classical Vaughan–Williams classification and is therefore categorized as an “other anti-arrhythmic drug”.
- “Other anti-arrhythmic drugs” are sometimes referred to as Class V.
Classification:
- Class V – Other anti-arrhythmic drugs
- Digoxin – stimulates the vagus nerve.
- Vernakalant – acts selectively only on atrial myocardium -

Mechanism:
- Acts selectively only on atrial myocardium
- Reduces excitability, automaticity, and suppresses re-entry in atrial myocardium
- because it inhibits K⁺ channels
- Prolongs the non-nodal action potential (AP) and effective refractory period (ERP)
- Slows conduction velocity and reduces atrial excitability
- because it inhibits Na⁺ channels
- Is use-dependent (effect is stronger at higher heart rates)
- The effect is present at heart rate > 90/min.
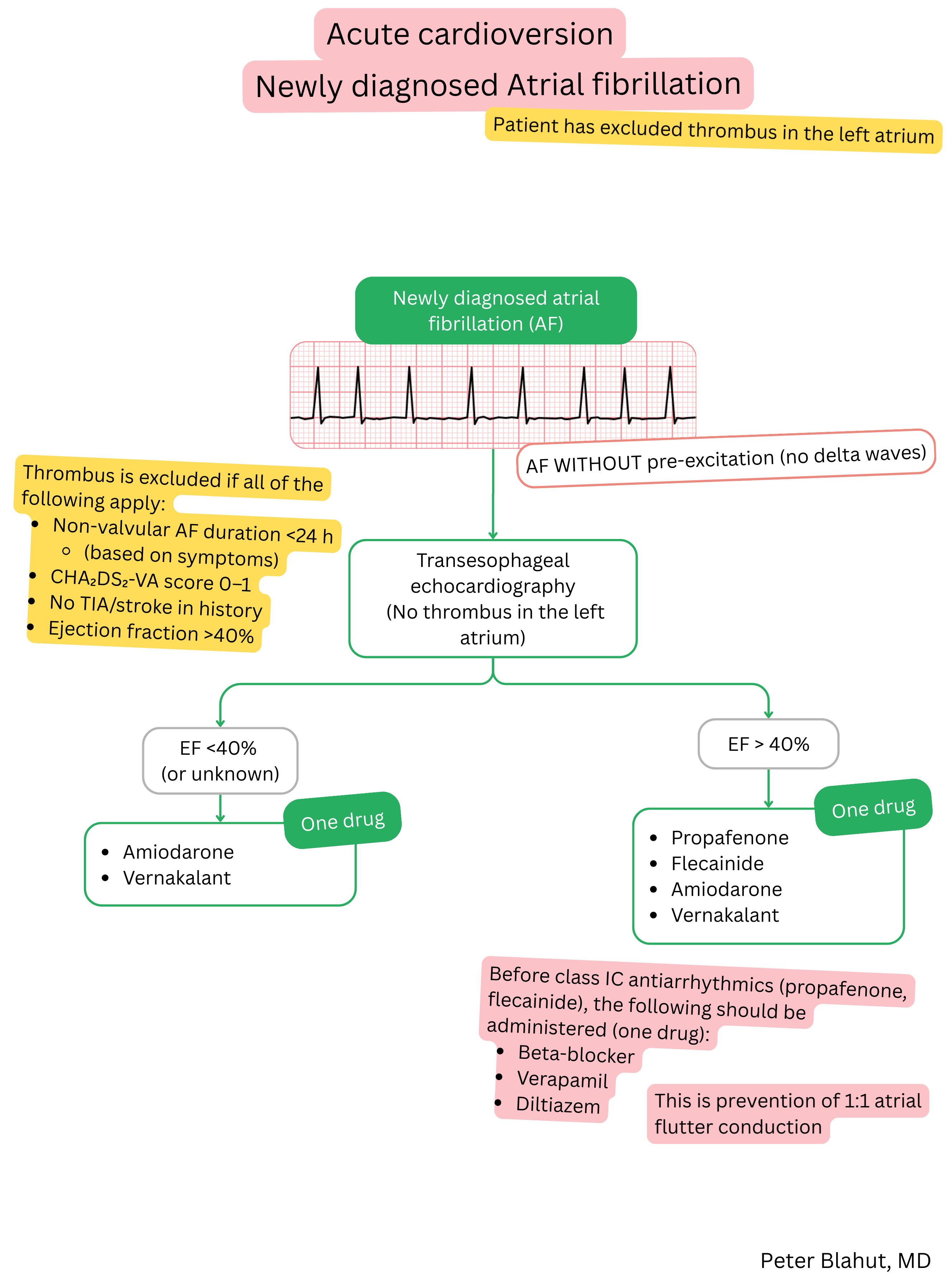
Effect on AF:
- Cardioversion of atrial fibrillation (AF) to sinus rhythm – termination of the AF episode and restoration of sinus rhythm
| Vernakalant and atrial fibrillation (AF) |
| Brand name |
| Brinavess |
| Indications |
- Acute cardioversion of AF to sinus rhythm
|
| Dosing |
- Acute cardioversion of AF to sinus rhythm (intravenous)
- 3 mg/kg over 10 minutes (maximum dose 339 mg)
- A second dose of 2 mg/kg over 10 minutes may be administered after 15 minutes if conversion to sinus rhythm has not occurred (maximum dose 226 mg)
- If conversion occurs during the first or second dose, the current infusion is completed.
|
| Onset of action |
- < 10 minutes (intravenous)
|
| Effect |
Time to conversion to sinus rhythm and success rate
- < 10 minutes – 50–70 % (intravenous)
|
| Duration of action |
|
|
| Contraindications |
- Severe aortic stenosis
- Hypotension (systolic blood pressure < 100 mmHg)
- Heart failure (NYHA III–IV)
- Acute coronary syndrome (within the last 30 days)
- Acute cardiac decompensation (within the last 30 days)
- Prolonged QT interval (QTc > 440 ms)
- Long QT syndrome
- Bradycardia (< 50/min)
- Sick sinus syndrome (without pacemaker)
- AV block II or III degree (without pacemaker)
- Intravenous Class I or III anti-arrhythmic drugs administered 4 hours before or after vernakalant
- Allergy to vernakalant
|
Patient monitoring during administration of vernakalant:
- Stop if a reason for infusion interruption occurs (see table below).
| Patient monitoring during administration of vernakalant |
| Monitoring time |
What to monitor |
Reason for infusion interruption |
| During infusion (0–10 min.) |
ECG monitoring (QTc interval)
Blood pressure
|
QTc > 500 ms
Torsades de pointes
Bradycardia < 40/min
Hypotension < 90/60 mmHg
|
| 30–120 min. after administration |
ECG
Blood pressure
|
Atrial flutter
QTc > 500 ms
Arrhythmias
|
Adverse effects:
- Very common (> 10 %)
- Hypotension
- Taste disturbance
- Sneezing
- Common (1–10 %)
- Atrial flutter
- Bradycardia
- Ventricular arrhythmia
- Hypertension
- AV block I degree
- Paresthesia
- Dizziness
- Fatigue
- Sensation of warmth
- Sweating
- Pruritus
- Vomiting
- Diarrhoea
- Uncommon (< 1 %)
- QRS widening on ECG
- QT interval prolongation
- AV block II or III degree
- Angina pectoris
Vernakalant and ibutilide are intravenous anti-arrhythmic drugs used for pharmacological cardioversion of AF.
- They belong to different classes and their main properties differ in part.
| Ibutilide vs. vernakalant in atrial fibrillation (AF) |
| Characteristic |
Ibutilide |
Vernakalant |
| Class |
Class III – K+ channel blocker |
“Other anti-arrhythmic drug” (blocks Na+ and K+ channels) |
| Mechanism of action |
Acts on atria, ventricles, and accessory pathways |
Acts only on atria |
| Indication |
Acute intravenous cardioversion of AF and flutter |
Acute intravenous cardioversion of AF |
| Use in pre-excited AF |
Yes |
Contraindicated |
| Use in atrial flutter |
Yes |
No |
| Success rate of conversion to sinus rhythm |
~30–50 % (AF), ~60–75 % (flutter) |
~50–70 % (AF) |
| Adverse effects |
QT interval prolongation, torsades de pointes |
Hypotension, bradycardia, dysgeusia, paraesthesia |