

Atrial remodelling is a consequence of atrial cardiomyopathy,
In remodelled atria, electrical changes develop that create the substrate for atrial fibrillation (AF).
Electrical changes arise mainly at sites where one anatomical structure transitions into another:
An episode of AF requires two components for initiation and maintenance:

Trigger
Substrate
The initiation and duration of an AF episode depend on the complex interaction between trigger, substrate, and autonomic modulation.
Which premature atrial contraction or salvo activates the substrate and how long the AF episode persists is extremely variable. It depends mainly on:
In paroxysmal AF, the trigger predominates
In persistent AF, the substrate predominates
Trigger and substrate
AF begins as paroxysmal; episodes of AF usually terminate spontaneously within 24 hours.
90% of paroxysmal AF has its trigger (usually also the substrate) in the region of the pulmonary vein ostia.
| Triggers of Atrial Fibrillation (Localization) | ||
|---|---|---|
| Anatomical site | Basic anatomy | Prevalence (%) |
| Pulmonary vein ostia | Myocardial sleeves extending 1–4 cm into the pulmonary veins (especially the superior pulmonary veins) | 85–90 % |
| Posterior wall of the left atrium | The posterior wall of the left atrium and the pulmonary vein antra share a common embryological origin. This tissue has different electrophysiological properties compared with the remaining atrial myocardium. | 5–10 % |
| Superior vena cava | Myocardial sleeves at the junction of the right atrium and the superior vena cava | 2–5 % |
| Crista terminalis | Ridge between the smooth and trabeculated parts of the right atrium | 1–3 % |
| Ligament of Marshall | Remnant of the embryonic left superior vena cava; an epicardial tract connecting the coronary sinus with the region of the left pulmonary veins. | 1–3 % |
| Ostium of the coronary sinus | Myocardial sleeves at the transition of the coronary sinus into the right atrium. | 1–3 % |
| Left atrial appendage | A small pouch located anterolaterally on the left atrium. The standard appendage volume is 5–10 ml; in atrial fibrillation 10–20 ml. | 1–3 % |
90% of paroxysmal AF has a trigger, often in combination with a substrate, localized in the region of the pulmonary vein ostia.
| Triggers in the Pulmonary Veins in Atrial Fibrillation (Localization) | ||
|---|---|---|
| Pulmonary vein | Prevalence | Note |
| Left superior | 45–50 % | The most frequent and most aggressive trigger source. Often shares a common ostium (carina) with the left inferior pulmonary vein. |
| Right superior | 30–35 % | Second most frequent source. Anatomical proximity to the sinus node and the superior vena cava. |
| Left inferior | 10–15 % | Common trigger in the presence of the anatomical variant “common trunk”. |
| Right inferior | 5–10 % | Least frequent trigger source |
Anatomical variants of the pulmonary veins are common and represent an important factor in planning AF ablation.
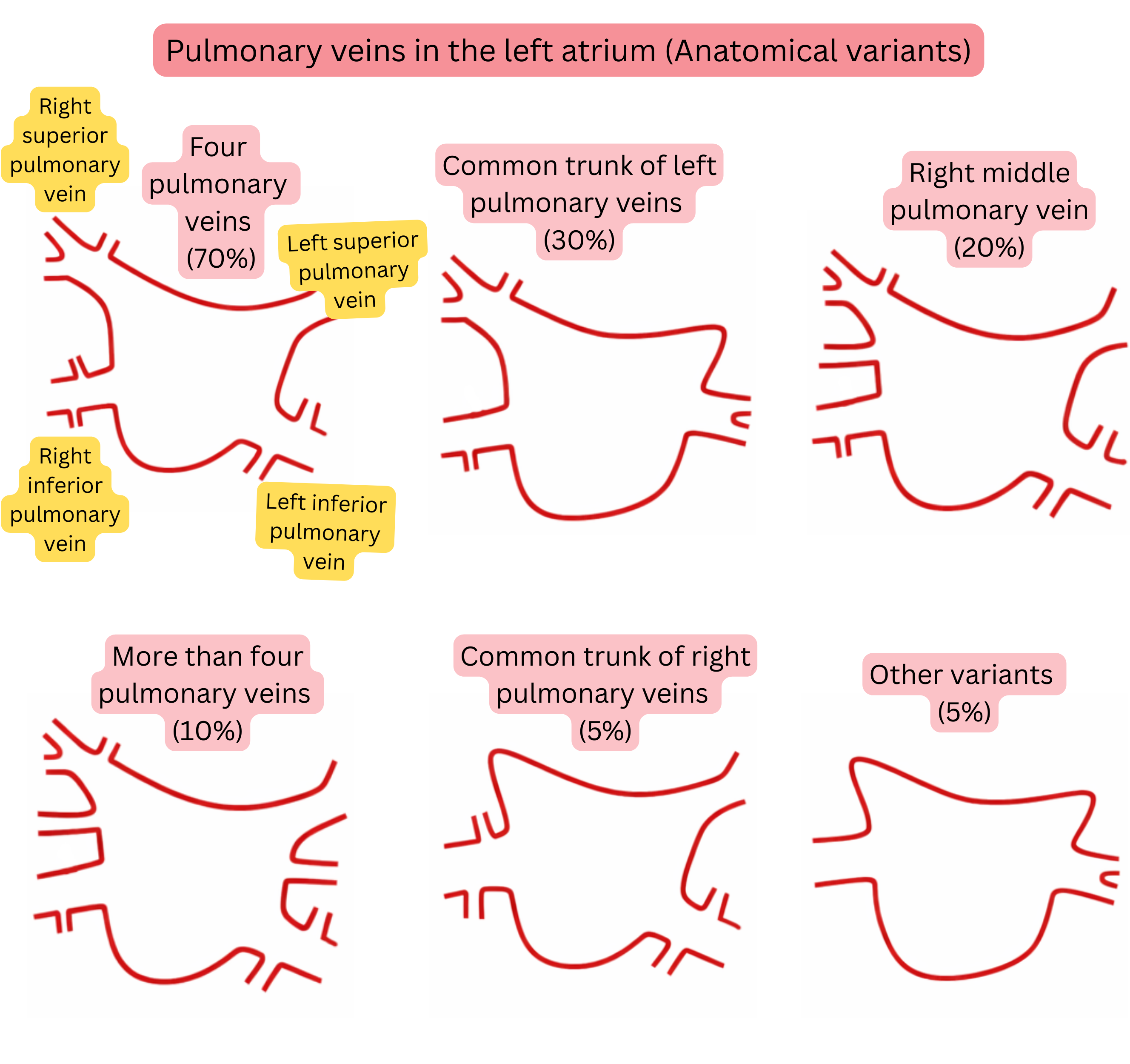
| Anatomical Variants of Pulmonary Veins in the Left Atrium | |
|---|---|
| Anatomical variant | Prevalence |
| Typical anatomy (4 separate pulmonary veins) | 60–70 % |
| Common trunk of the left pulmonary veins | 20–30 % |
| Right middle pulmonary vein (accessory) | 15–25 % |
| More than 4 pulmonary veins (accessory) | 5–10 % |
| Common trunk of the right pulmonary veins | <5 % |
| Early bifurcation of the pulmonary veins | 10–15 % |
Trigger and substrate
| Substrate of Atrial Fibrillation (Localization) | ||
|---|---|---|
| Anatomical site | Basic anatomy | Prevalence (%) |
| Posterior wall of the left atrium | Bounded by the pulmonary veins. This is the most frequent site of fibrosis. | 60–70 % |
| Pulmonary vein ostia | Myocardial sleeves extending 1–4 cm into the pulmonary veins (especially the superior pulmonary veins) | 50–60 % |
| Roof of the left atrium | Region connecting the superior pulmonary veins. | 30–40 % |
| Interatrial septum | Region around the fossa ovalis and Bachmann’s bundle. | 20–30 % |
| Left atrial appendage | The trabeculated musculature of the appendage may represent a substrate. | 10–20 % |
| Right atrium | Substrate according to prevalence:
|
10–20 % |
| Mitral isthmus | Zone between the left inferior pulmonary vein and the mitral annulus. Critical for the development of perimitral flutter. 30–50% of atypical left atrial flutters pass through the mitral isthmus. | 10–20 % |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
