

The interest of arrhythmology in structural heart disease and Class IC antiarrhythmic drugs
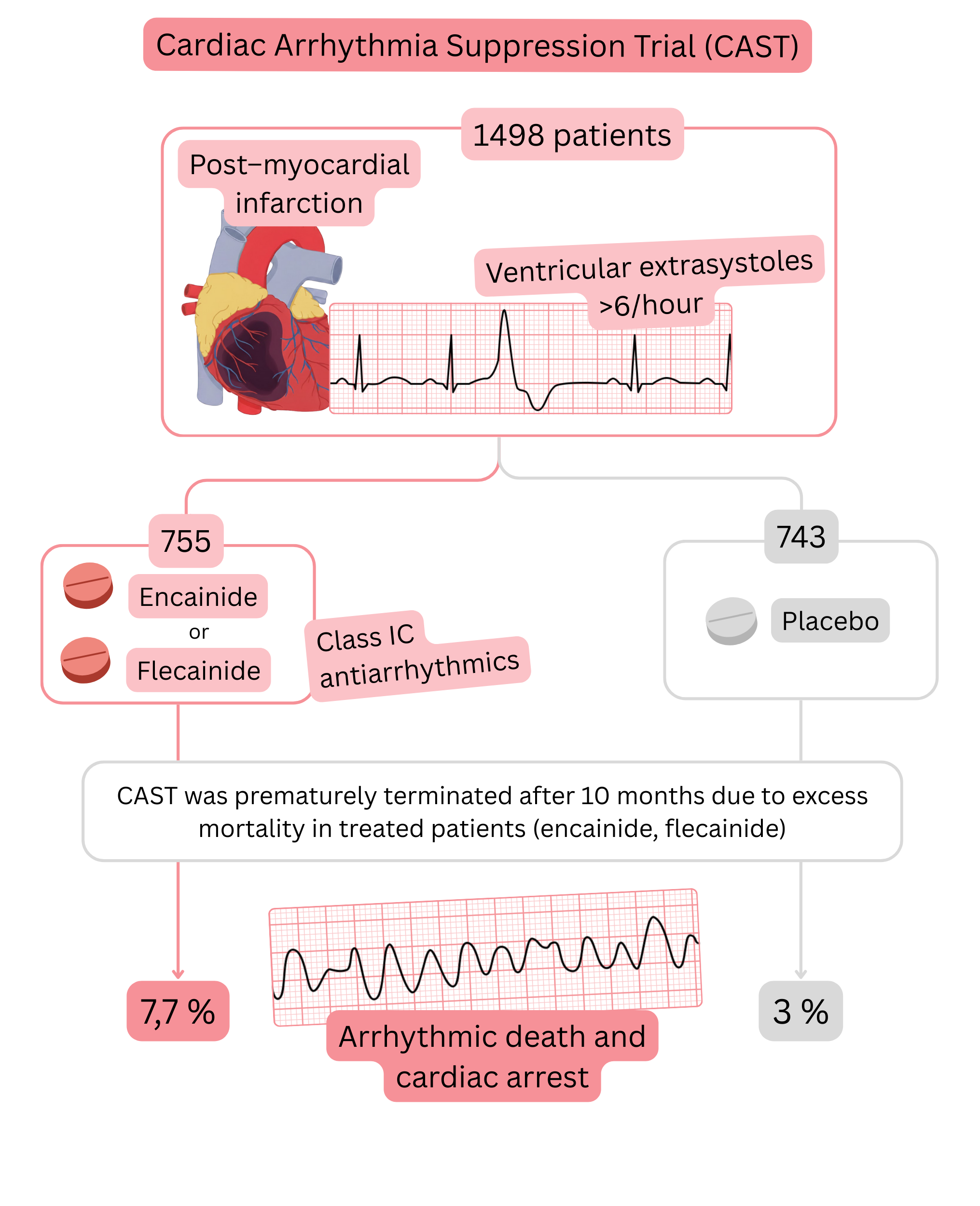
CAST trial
Summary of the CAST trial:
Mechanism of ventricular arrhythmia in the CAST trial
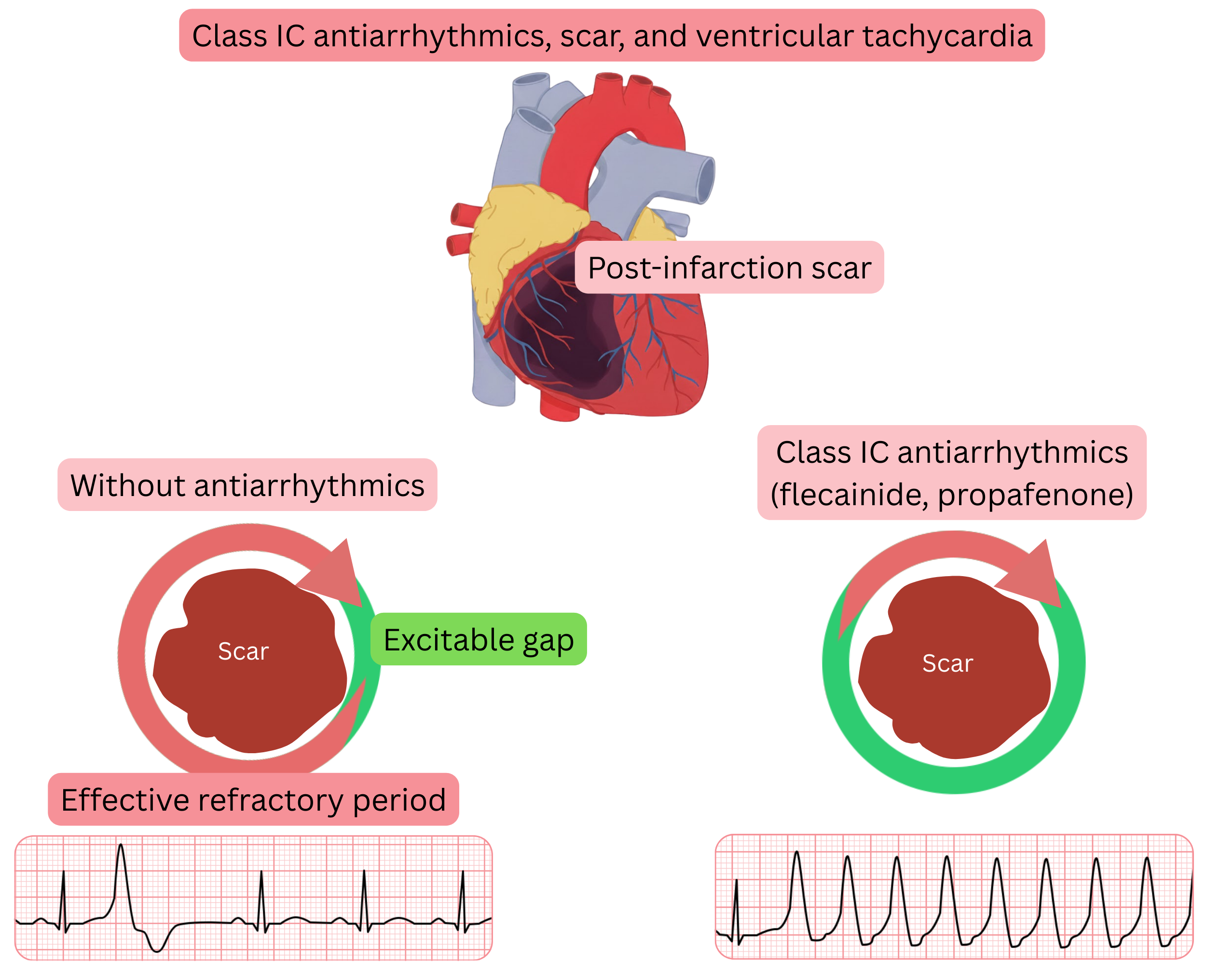
Extension of the CAST trial conclusions into clinical practice
Structural heart disease
| Structural heart disease |
|---|
| Previous myocardial infarction |
| Coronary artery disease |
| Ejection fraction (<40 %) |
| Left ventricular hypertrophy (>15 mm) |
| Cardiomyopathy (dilated, hypertrophic, restrictive, infiltrative) |
| Valvular disease – stenosis or regurgitation (moderate or severe) |
| Heart failure (NYHA II–IV, hospitalization for heart failure) |
| Status post cardiac surgery |
In the following table, you can review the basic diagnostic methods and parameters used to diagnose structural heart disease.
| Structural heart disease (diagnostics) | |
|---|---|
| Diagnosis | Diagnostics |
| Previous myocardial infarction |
ECG: pathological Q waves (≥ 40 ms, ≥ 25 % of QRS, ≥ 2 leads) Echo: regional wall motion abnormality (akinesia, dyskinesia) MRI: scar (LGE positive finding) |
| Coronary artery disease (IHD) |
CT coronary angiography: stenosis > 50 % left main, > 70 % other major branches Exercise test: ST depression > 1 mm during stress = ischaemia |
| Ejection fraction (< 40 %) |
Echo: EF < 40 % MRI: EF < 40 % |
| Left ventricular hypertrophy |
ECG: Sokolow–Lyon index > 35 mm Echo: wall thickness > 15 mm |
| Cardiomyopathy |
Dilated: LVEDD > 55 mm + EF < 40 % (echo/MRI) Hypertrophic: LV wall ≥ 15 mm (echo/MRI) Restrictive: biatrial dilatation + diastolic dysfunction (E/e´ > 15) Infiltrative: echo – speckled myocardium; MRI – diffuse LGE |
| Valvular disease (stenosis / regurgitation) | Echo: stenosis or regurgitation (moderate or severe) |
| Heart failure (NYHA II–IV) |
Clinical: dyspnoea on exertion or at rest, oedema, orthopnoea, recurrent hospitalizations Echo: EF < 40 % (HFrEF) or significant diastolic dysfunction (HFpEF / HFmrEF) BNP > 35 pg/ml or NT-proBNP > 125 pg/ml |
| Status post cardiac surgery | History: documented surgery (CABG, valve surgery, congenital defect) |
In the following table, you can review the investigations and criteria that must be fulfilled for the safe administration of Class IC antiarrhythmic drugs in patients with atrial fibrillation (AF).
| Criteria for administration of Class IC antiarrhythmic drugs in atrial fibrillation | |
|---|---|
| Investigation | Criteria |
| ECG |
No pathological Q waves (≥ 40 ms, ≥ 25 % of QRS, ≥ 2 leads) No hypertrophy: Sokolow–Lyon index ≤ 35 mm (S in V1 + R in V5 or V6) QRS < 120 ms (no bundle branch block) QTc by sex: men < 450 ms, women < 470 ms |
| Echocardiography |
Ejection fraction (EF) ≥ 40 % Wall thickness ≤ 15 mm No dilatation: LVEDD < 55 mm, LA < 40 mm or < 34 ml/m² Valves: at most mild regurgitation or stenosis |
| Exercise testing |
Negative for ischaemia No induced arrhythmias No drop in systolic blood pressure > 10 mmHg during exercise |
| CAG / CTA / MRI |
Indicated in case of clinical suspicion of IHD or cardiomyopathy Significant coronary artery stenosis, post-infarction scar, or cardiomyopathy must be excluded |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
