

Before atrial fibrillation (AF) ablation, 4 weeks of anticoagulation therapy is recommended as prevention of thromboembolism.
For prevention of thromboembolism, 2 months of anticoagulation therapy is recommended after ablation,
Transoesophageal echocardiography before AF ablation (<24 h) is recommended despite 4 weeks of anticoagulation therapy in the following cases:
| TEE before ablation – indications despite anticoagulation (4 weeks) |
|---|
| History of transient ischaemic attack (TIA) |
| History of stroke |
| Irregular use of anticoagulation therapy |
| INR < 2 (during warfarin therapy) |
| History of intracardiac thrombus (especially in the left atrial appendage) |
| History of left atrial appendage emptying velocity < 20 cm/s |
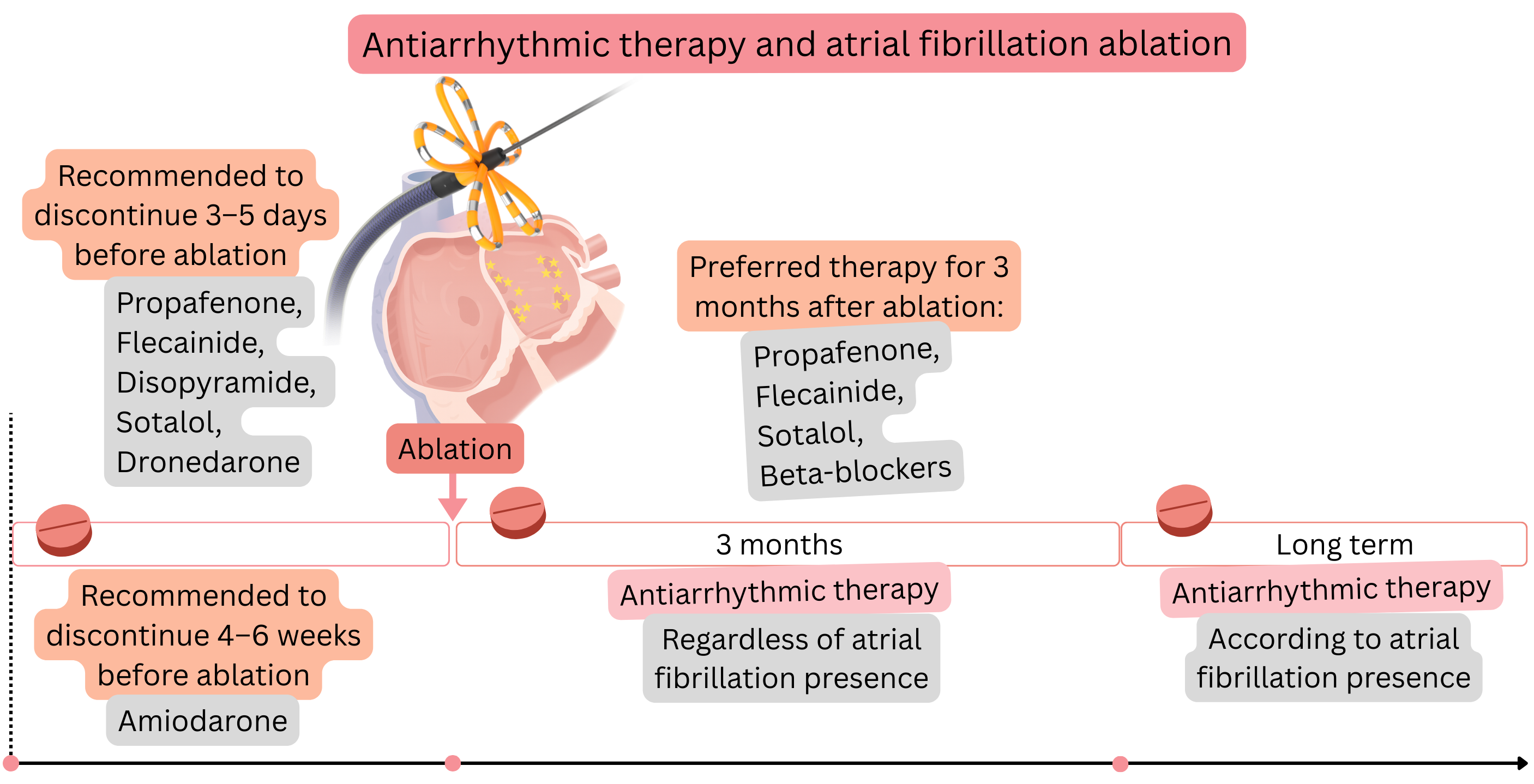
Before AF ablation, it is appropriate to discontinue anti-arrhythmic therapy (if the patient’s condition allows).
Before ablation, it is appropriate to discontinue anti-arrhythmic drugs used for rhythm control (if the patient’s condition allows), but not those used for rate control.
| Discontinuation of anti-arrhythmic drugs before atrial fibrillation ablation | ||
|---|---|---|
| Drug | Class | Discontinuation before ablation |
| Disopyramide | IA | 3–5 days |
| Propafenone | IC | 3–5 days |
| Flecainide | IC | 3–5 days |
| Sotalol | III | 3–5 days |
| Amiodarone | III | 4–6 weeks |
| Dronedarone | III | 3–5 days |
AF ablation (procedure) requires 3 vascular access sheaths via the groins (femoral veins):
After insertion of the femoral sheaths, 5000 IU of unfractionated heparin (UFH) is administered.
| UFH dose and ACT prolongation | ||
|---|---|---|
| UFH dose | ACT at 70 kg (prolongation from normal) | ACT at 100 kg (prolongation from normal) |
| No UFH | 80 – 120 s | 80 – 120 s |
| 1000 IU | 120 – 140 s (↑20–40) | 110 – 130 s (↑10–30) |
| 3000 IU | 150 – 180 s (↑50–80) | 130 – 160 s (↑30–60) |
| 5000 IU | 200 – 240 s (↑100–140) | 170 – 210 s (↑70–110) |
| 7000 IU | 230 – 280 s (↑130–180) | 200 – 250 s (↑100–150) |
| 10000 IU | 280 – 340 s (↑180–240) | 230 – 300 s (↑130–200) |
A thrombus in the region of the venous sheaths and in the right atrium on the catheter is not as dangerous, as it embolizes to the lungs.
| Risk of thrombus formation on the sheath or catheter | |
|---|---|
| ACT | Estimated risk |
| 80 – 120 s (no UFH) | 10–20 % (within 10–20 min.) |
| 250–300 s | 1–2 % |
| 300–350 s | < 1 % |
After introducing a dedicated needle via the femoral vein into the right atrium, a transseptal puncture is performed.
ACT and UFH during the procedure
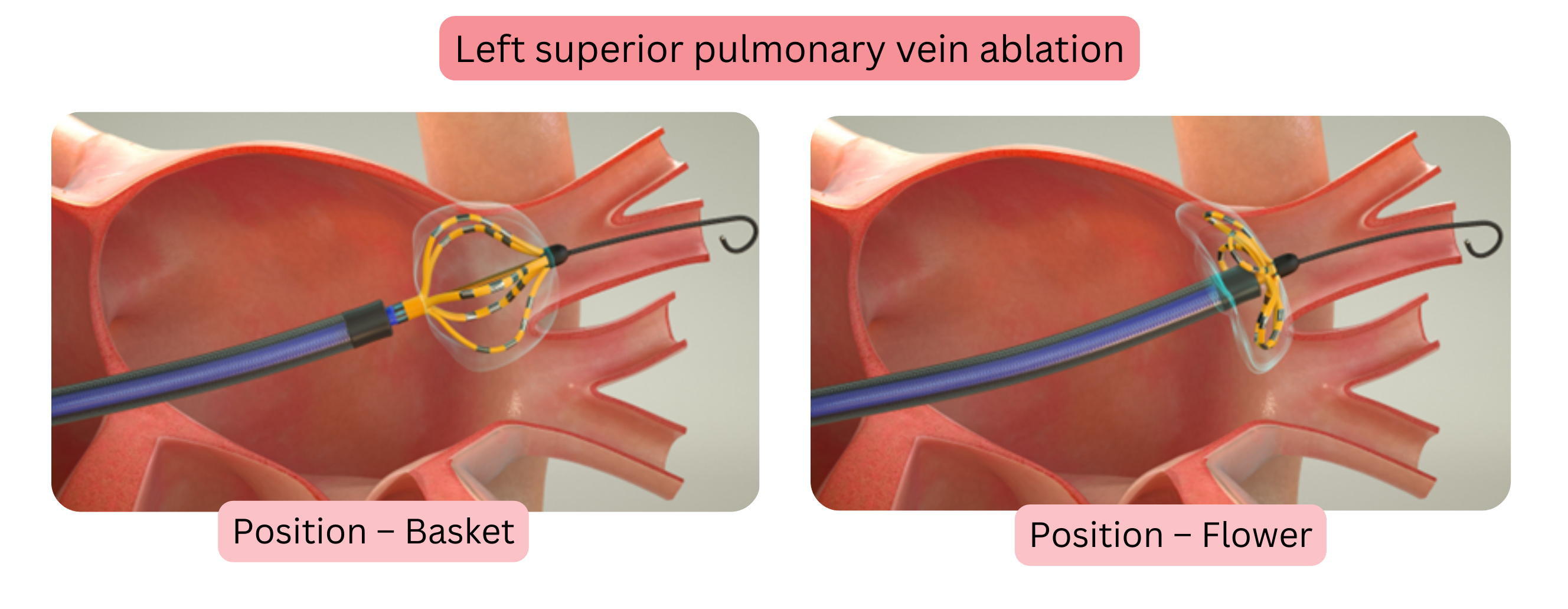
Farawave is a dedicated catheter with two configurations: basket and flower.
3–5 min before the first application, 1 mg atropine is administered intravenously (may be repeated up to a maximum dose of 3 mg).
| Atropine – prevention of bradycardia before ablation | |
|---|---|
| Purpose | Prevention of bradycardia |
| Mechanism | Blocks vagal effect (antimuscarinic effect) |
| Dose | 1 mg intravenous (max. 3 mg) |
| Onset of action | 1–2 min |
| Expected effect | Heart rate increase by 20–40/min |
| Duration of action | 30–60 min |
| Contraindication | Glaucoma |
Electrical pulses are painful; therefore, they are delivered under general anaesthesia or sedation.
If AF persists after pulmonary vein isolation, a more extensive left atrial ablation is performed:
Left atrial appendage and ablation.
If a patient has AF and atrial flutter (AFL),
During pulsing in AF and AFL ablation, coronary spasm may occur if ablation is performed near a coronary artery.
| Prevention of coronary spasm before ablation – Isoket vs. nitroglycerin | ||
|---|---|---|
| Drug | Isoket (isosorbide dinitrate) | Nitroglycerin |
| Purpose | Slower, longer-lasting prevention of coronary spasm | Rapid prevention and treatment of coronary spasm |
| Mechanism of action | NO donor → coronary vasodilation | NO donor → coronary vasodilation |
| Administration | intravenous bolus 1–2 mg | intravenous bolus 50–200 µg |
| Onset of action | 3–5 min | 1–2 min |
| Duration of action | 30–60 min | 5–10 min |
| Risk of hypotension | Higher (longer duration) | Lower (short duration) |
ECG changes during ablation in the region of the mitral and tricuspid isthmus:
Electrical pulses in pulsed field ablation are cardioselective:
| Electrical pulses in pulsed field ablation |
|---|
| Do not cause pulmonary vein stenosis |
| Do not cause atrio-oesophageal fistula |
| Do not cause phrenic nerve injury |
After AF ablation, procedural success can be verified in three ways:
| Verification of atrial fibrillation ablation success | |||
|---|---|---|---|
| Method | Site of pacing | What is assessed | Success |
| Pacing from the pulmonary veins | Sequentially from each pulmonary vein (using the Farawave catheter) | Whether impulses from the vein propagate to the left atrium | No atrial response = vein isolated |
| Burst pacing | From the coronary sinus (CS catheter) | Induction of AF after bursts (8–10 stimuli) | AF is not induced or AF does not last > 1 min = success |
| Ramp pacing | From the coronary sinus (CS catheter) | Whether AF is induced during pacing with progressive cycle shortening | AF is not induced or AF does not last > 1 min = success |
During pulsed field ablation, electroporation of cardiomyocytes occurs, which is perforation of the cell membrane by an electric field. This is followed by apoptosis. Cardio-specific enzymes are released from damaged cardiomyocytes and rise to high levels within 24 h.
| Cardio-specific enzymes within 24 h after pulsed field ablation | |
|---|---|
| Enzyme | Value |
| Troponin | 1500 ng/l (±500) |
| CK | 300 ng/l (±100) |
| CK-MB | 35 mg/l (±10) |
During pulsed field ablation, haemolysis occurs, which manifests after the procedure as macroscopic haematuria.
Ablation success is assessed by the presence of AF recurrence within 1 year after ablation.
| Atrial fibrillation ablation success (within 12 months) | |
|---|---|
| AF classification | Success |
| Paroxysmal AF | 66–82 % |
| Persistent AF | 56–72 % |
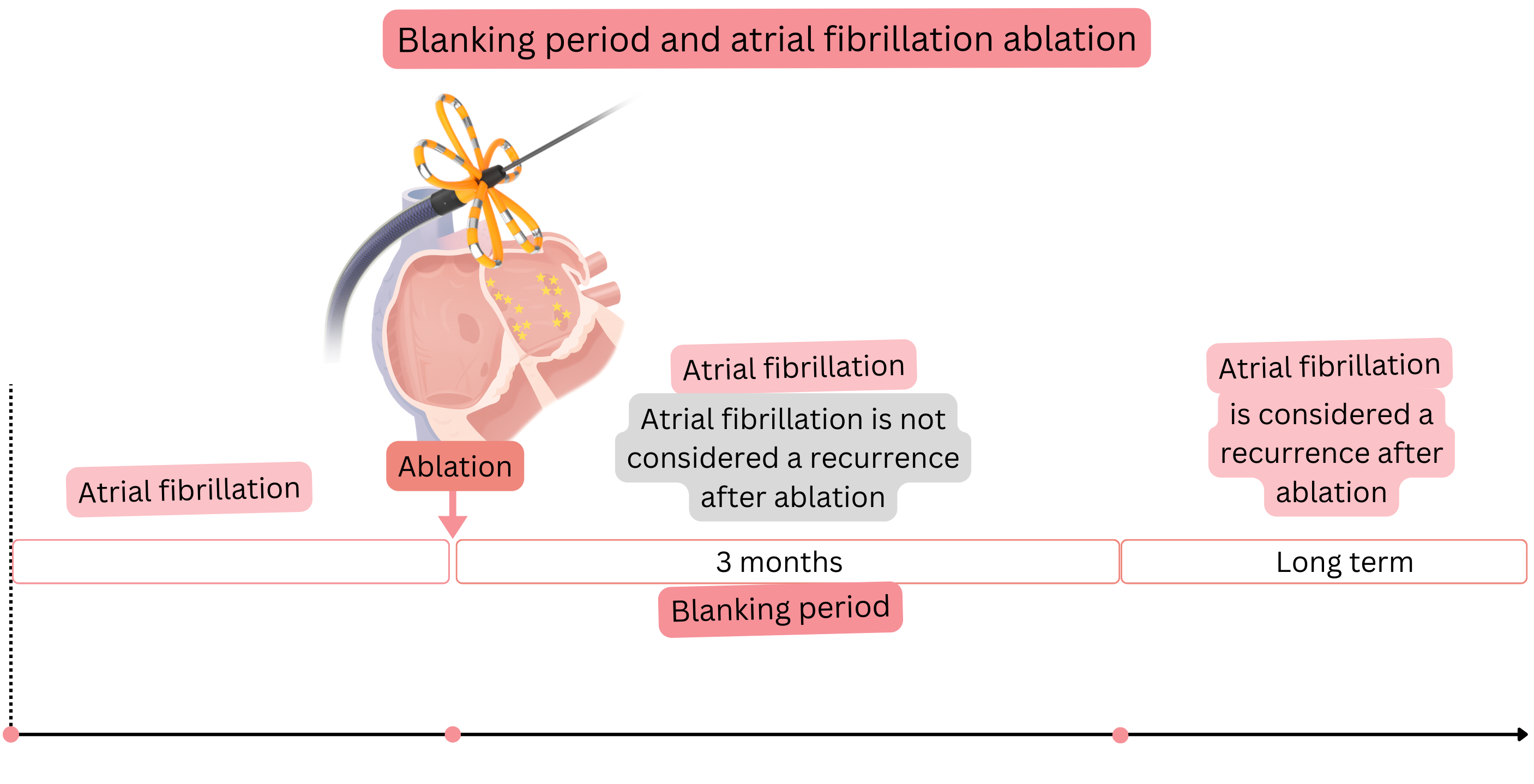
Blanking period
| Therapy during the blanking period (3 months after ablation) | ||
|---|---|---|
| Therapy | Duration | Note |
| Anti-arrhythmic drugs | 3 months | Administered regardless of whether sinus rhythm or AF is present. |
| Anticoagulation therapy | 2 months | Administered regardless of the CHA2DS2-VA score |
In AF recurrence after ablation—pulmonary vein isolation (with pulsed field energy), reconnection of a pulmonary vein to the left atrium may occur. Reconnection most commonly occurs in:
During and after pulsed field AF ablation, complications may occur, but they are very rare. The incidence of complications is:
Major and minor complications are listed in the following table:
| Major complications of pulsed field ablation | |
|---|---|
| Total | 0.98 % |
| Pericardial tamponade | 0.36 % |
| Vascular complication (requiring intervention) | 0.30 % |
| Coronary spasm | 0.14 % |
| Stroke | 0.12 % |
| Haemolysis with acute renal failure | 0.03 % |
| Death | 0.03 % |
| Other (thrombosis, coronary air embolism) | 0.006 % |
| Oesophageal fistula | 0 % |
| Pulmonary vein stenosis | 0 % |
| Phrenic nerve injury (permanent) | 0 % |
| Minor complications of pulsed field ablation | |
|---|---|
| Total | 3.21 % |
| Vascular complications (not requiring intervention) | 2.20 % |
| Pericardial effusion (not requiring intervention) | 0.33 % |
| Other minor complications (haematomas, arrhythmias) | 0.32 % |
| Pericarditis | 0.17 % |
| Transient ischaemic attack | 0.12 % |
| Phrenic nerve injury (temporary) | 0.06 % |
In 5–15% of patients, asymptomatic silent cerebral ischaemia occurs during pulsed field ablation.
| Atrial fibrillation ablation | Class |
|---|---|
| Pulsed field ablation (not radiofrequency or cryoablation) is recommended as the preferred method of atrial fibrillation ablation. | I |
Pulsed field ablation is recommended in patients with paroxysmal or persistent atrial fibrillation if atrial fibrillation is symptomatic:
|
I |
| Pulsed field ablation is recommended in patients with tachycardia-induced cardiomyopathy due to atrial fibrillation. | I |
| Pulsed field ablation should be considered in patients with atrial fibrillation who have symptomatic pre-automatic pauses. | IIa |
In atrial fibrillation recurrence, pulsed field ablation may be repeated (not earlier than 3 months) if atrial fibrillation is symptomatic:
|
IIa |
| Before atrial fibrillation ablation, CT or MR angiography of the left atrium and pulmonary veins should be considered to assess pulmonary vein anatomy. | IIa |
The “pace and ablate” strategy may be considered in patients with symptomatic atrial fibrillation in whom the following have failed:
|
IIa |
| Anticoagulation therapy and atrial fibrillation ablation | Class |
|---|---|
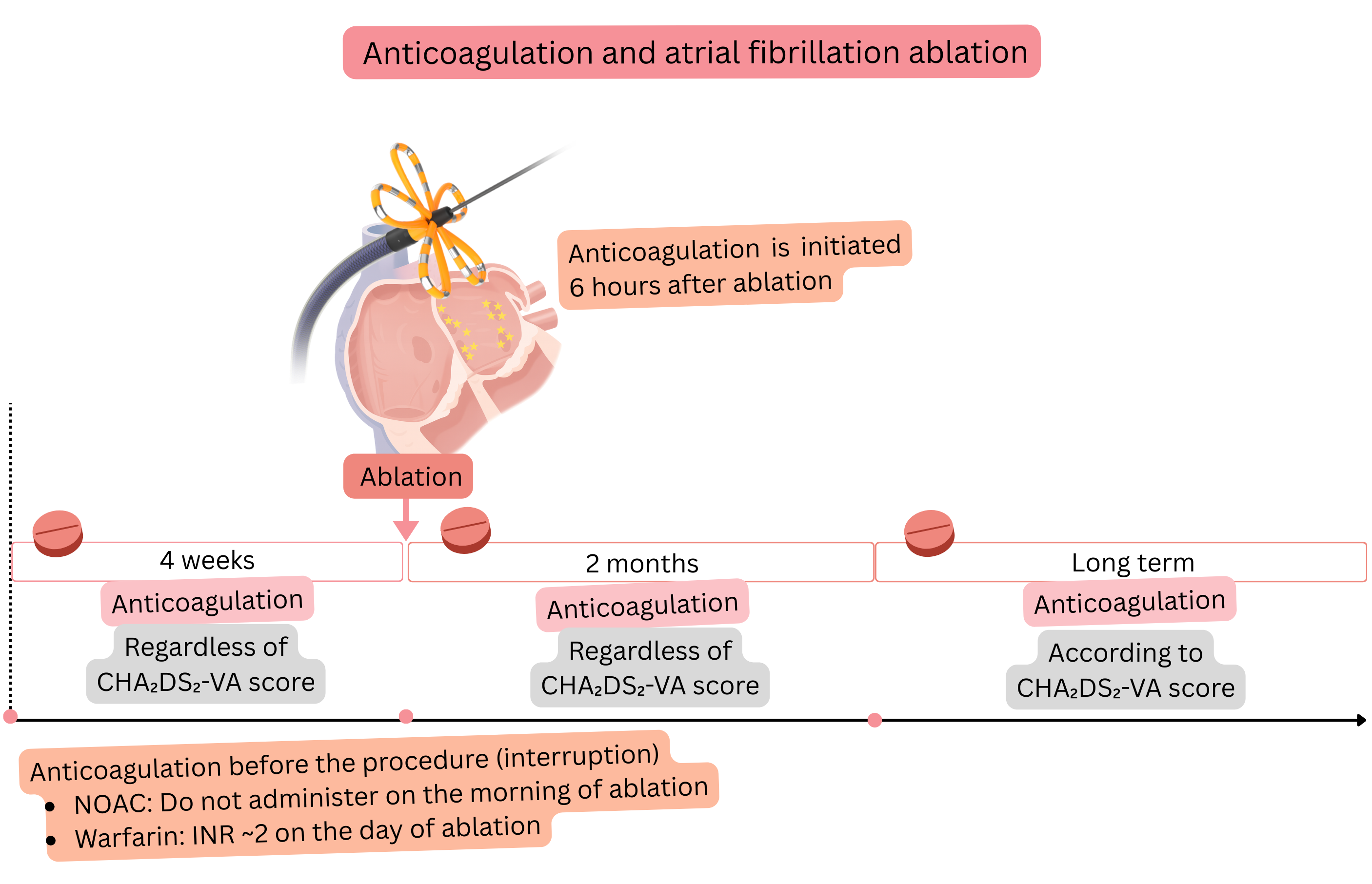
| Anticoagulation therapy is recommended for at least 4 weeks before atrial fibrillation ablation, regardless of the CHA₂DS₂-VA score. | I |
| NOAC anticoagulation therapy is recommended not to be taken in the morning on the day of atrial fibrillation ablation. | I |
| NOAC anticoagulation therapy is recommended to be started 6 h after atrial fibrillation ablation if there are no signs of bleeding. | I |
| During warfarin therapy, atrial fibrillation ablation is recommended to be performed with a therapeutic INR of approximately 2.0 on the day of the procedure. | I |
| Anticoagulation therapy is recommended for the first 2 months after atrial fibrillation ablation, regardless of ablation success and regardless of the CHA₂DS₂-VA score. | I |
| Two months after atrial fibrillation ablation, long-term anticoagulation is indicated according to the CHA₂DS₂-VA score, regardless of ablation success. | I |
| Anti-arrhythmic therapy (propafenone, flecainide, sotalol, beta-blockers) is recommended for the first 3 months after atrial fibrillation ablation, regardless of ablation success. | I |
| Three months after atrial fibrillation ablation, anti-arrhythmic therapy is indicated according to atrial fibrillation recurrence. | I |
| Atrial fibrillation ablation may be considered if the patient is receiving dual antithrombotic therapy (e.g. NOAC + clopidogrel). | IIa |
| Atrial fibrillation ablation during cardiac surgery | Class |
|---|---|
| In a patient undergoing cardiac surgery on the mitral valve, concomitant surgical atrial fibrillation ablation using the Cox-Maze IV procedure is recommended. | I |
| In a patient undergoing cardiac surgery other than mitral valve surgery, concomitant surgical atrial fibrillation ablation using the Cox-Maze IV procedure should be considered. | IIa |
| During cardiac surgery, the presence of thrombus in the left atrium is recommended to be excluded before surgical atrial fibrillation ablation. | I |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
