

Classification:
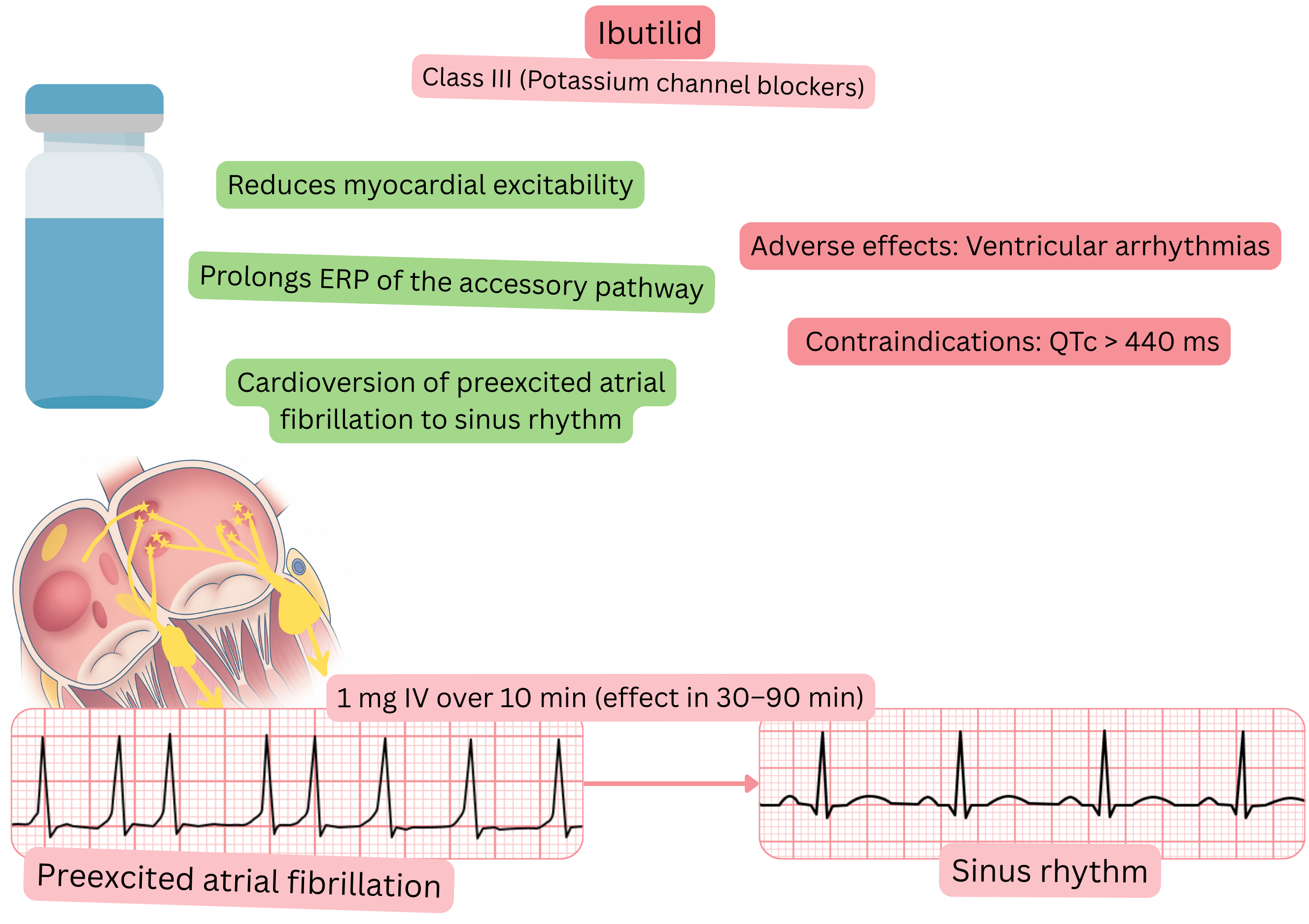
Mechanism:
Effect:
| Ibutilide and atrial fibrillation (AF) |
|---|
| Brand name |
| Convert |
| Indications |
|
| Dosing |
|
| Onset of action |
|
| Effect |
Time to conversion to sinus rhythm and success rate
|
| Duration of action |
|
| Contraindications |
|
Patient monitoring during ibutilide administration:
| Patient monitoring during ibutilide administration | ||
|---|---|---|
| Monitoring period | What to monitor | Reason for discontinuation |
| During infusion (0–10 minutes) |
ECG (QTc interval) Blood pressure |
QTc > 500 ms Torsades de pointes Bradycardia < 40/min. Hypotension < 90/60 mmHg |
| 30–120 minutes after administration |
ECG Blood pressure |
QTc > 500 ms Arrhythmias |
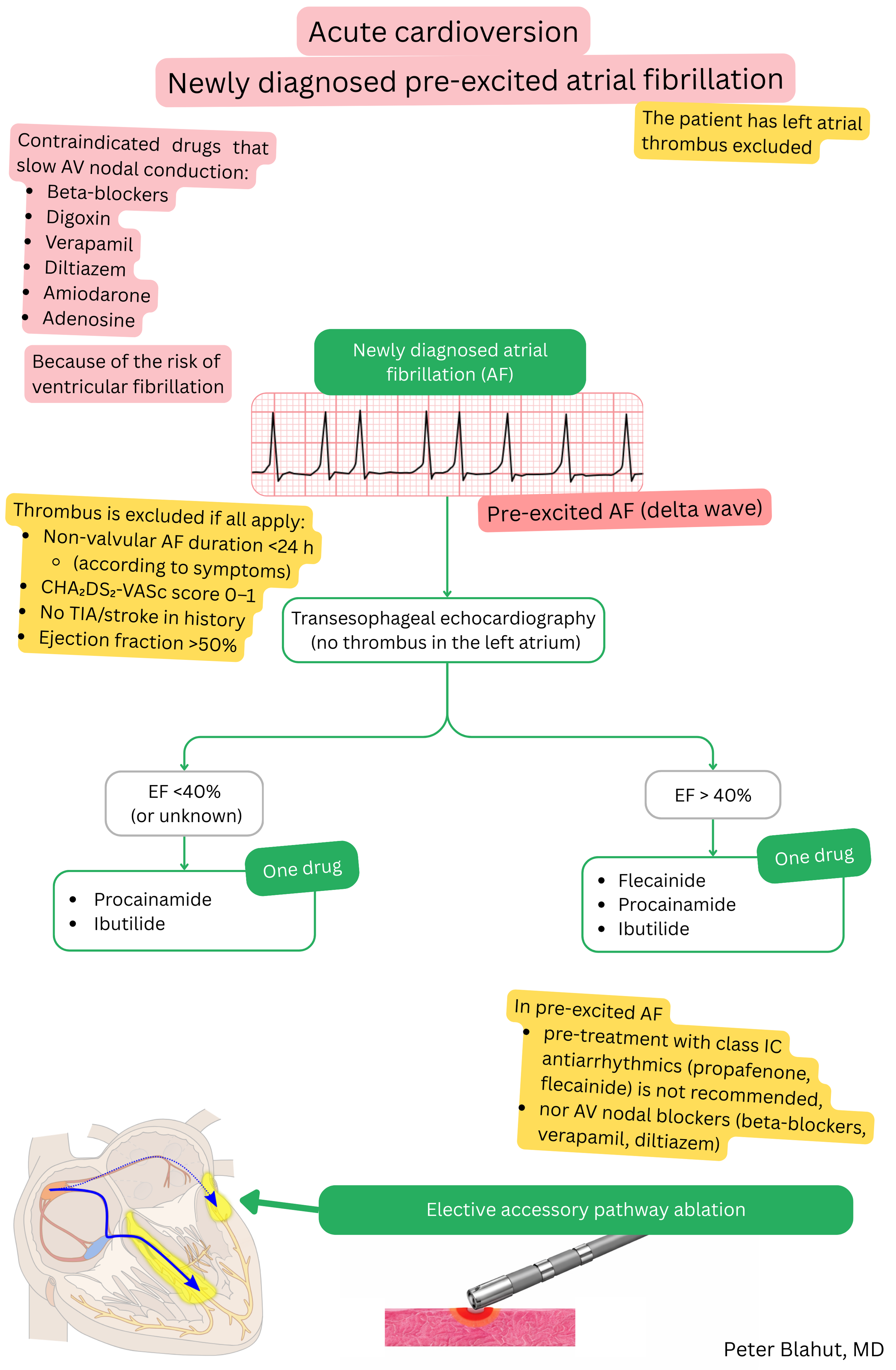
Adverse effects:
Vernakalant and ibutilide are intravenous anti-arrhythmic drugs indicated for pharmacological cardioversion of AF.
| Ibutilide vs vernakalant and atrial fibrillation | ||
|---|---|---|
| Property | Ibutilide | Vernakalant |
| Class | Class III – K⁺ channel blocker | “Other anti-arrhythmic” (blocks Na⁺ and K⁺ channels) |
| Mechanism of action | Acts on atria, ventricles, and accessory pathways | Acts only on atria |
| Indication | Acute intravenous cardioversion of AF and flutter | Acute intravenous cardioversion of AF |
| Use in pre-excited AF | Yes | Contraindicated |
| Use in atrial flutter | Yes | No |
| Conversion success rate to sinus rhythm | ~30–50 % (AF), ~60–75 % (AFL) | ~50–70 % (AF) |
| Adverse effects | QT prolongation, torsades de pointes | Hypotension, bradycardia, dysgeusia, paraesthesia |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
