

Classification:
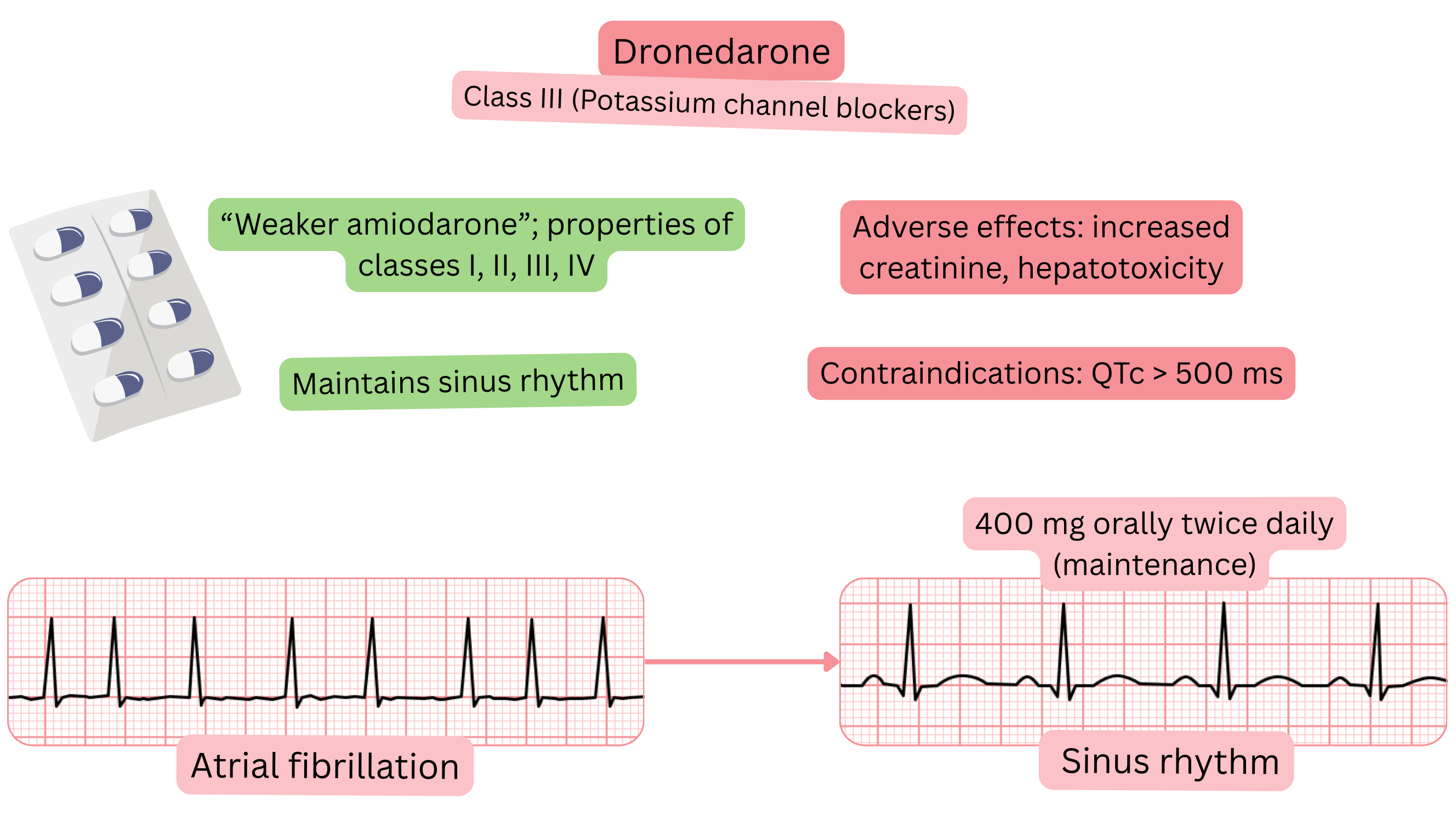
Mechanism:
Effect on AF:
| Dronedarone and atrial fibrillation (AF) |
|---|
| Brand name |
| Multaq |
| Indications |
|
| Dosing |
|
| Onset of action |
|
| Effect |
Maintenance of sinus rhythm (paroxysmal or persistent AF) at 1 year
|
| Duration of action |
|
| Contraindications |
|
Patient monitoring after initiation of dronedarone:
| Patient monitoring before and during dronedarone therapy | ||
|---|---|---|
| Time | What to monitor | Reason for discontinuation |
| Before initiation |
ECG (QTc interval, AV block) Liver tests (ALT, AST, GGT, ALP, bilirubin) |
QTc ≥ 500 ms Second- or third-degree AV block Abnormal liver tests |
| 2 months |
Liver tests ECG |
Marked deterioration of liver tests QTc interval ≥ 500 ms PR interval > 280 ms |
| 4 months |
Liver tests ECG |
Marked deterioration of liver tests QTc interval ≥ 500 ms PR interval > 280 ms |
| 6 months |
Liver tests ECG |
Marked deterioration of liver tests QTc interval ≥ 500 ms PR interval > 280 ms |
| Every 1 year |
Liver tests ECG Echocardiography |
Marked deterioration of liver tests QTc interval ≥ 500 ms PR interval > 280 ms Ejection fraction < 40% |
Adverse effects:
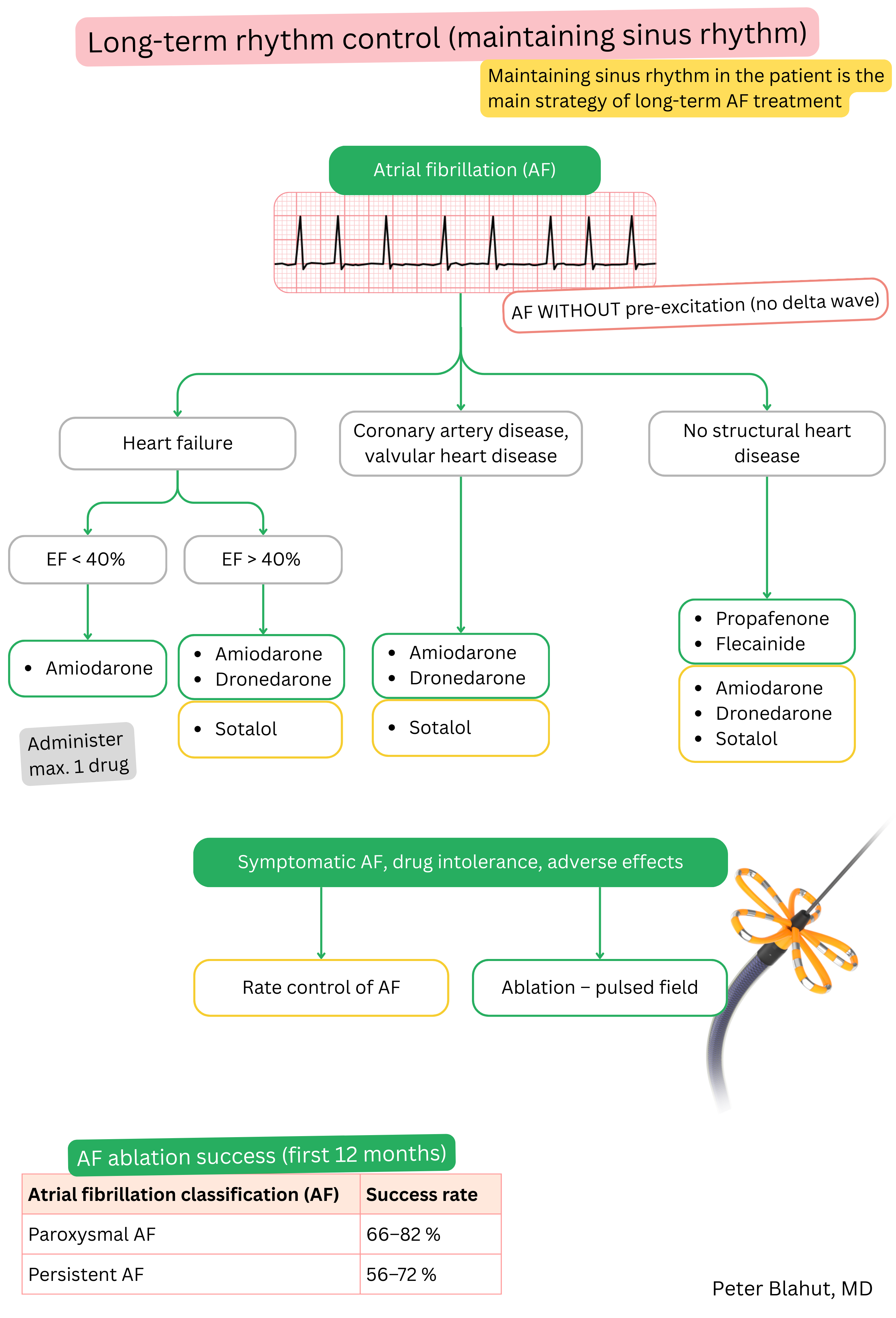
Amiodarone and dronedarone both belong to Class III anti-arrhythmic drugs but differ in their properties.
| Amiodarone vs dronedarone and atrial fibrillation | ||
|---|---|---|
| Property | Amiodarone | Dronedarone |
| Efficacy (maintenance of SR) | 60–70 % at 1 year | 30–40 % at 1 year |
| Onset of action | Slow (days–weeks, full effect after loading of ~10 g) | Faster (3–6 hours) |
| Duration of action | Persists 2–3 months after discontinuation | 12–24 hours (disappears after dose omission) |
| Suitable patient | Also with structural heart disease | Patient without structural heart disease and with preserved ejection fraction |
| Heart failure | May be used (including HFrEF) | Contraindicated (NYHA III–IV, HFrEF < 40 %) |
| Tissue accumulation | Yes – adipose tissue, lungs, eye, thyroid gland | Minimal |
| Lungs (toxicity) | Pulmonary fibrosis, interstitial pneumonitis | No pulmonary toxicity |
| Liver (toxicity) | Mild hepatotoxicity, elevated liver enzymes | Possible severe hepatitis, hepatic failure |
| Thyroid (toxicity) | Hypothyroidism and hyperthyroidism | No effect on the thyroid gland |
| Eyes (toxicity) | Corneal deposits, optic neuropathy | No ocular toxicity |
| Skin (toxicity) | Photosensitivity, blue-grey skin discoloration | Skin rash, pruritus |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
