

Chronic coronary syndrome (CCS)
Acute coronary syndrome (ACS)
Type 1 myocardial infarction
Type 2 myocardial infarction
Revascularization is restoration of blood flow across a stenosis or occlusion of a coronary artery. Revascularization methods include:
ACS is an acute critical condition that may trigger or worsen an AF episode.
The incidence of new-onset AF within 24 hours after ACS is 2–23 %.
10–15 % of patients with AF undergo PCI for coronary artery disease.
Tachy-AF (ventricular rate >100/min) may cause type 2 myocardial infarction.
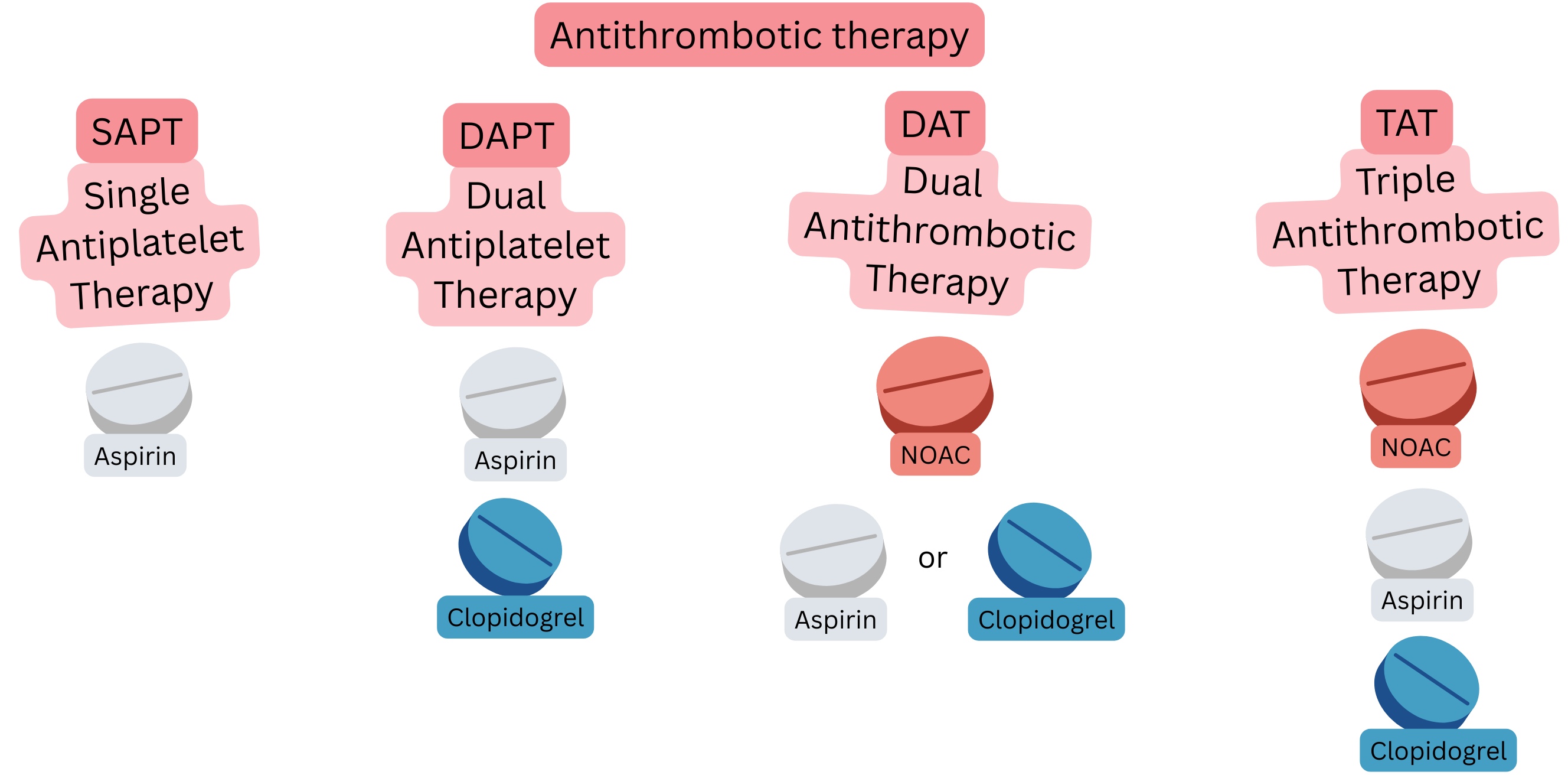
Patients with ACS and AF require combined antithrombotic therapy:
| Antithrombotic therapy (Terminology) in coronary syndrome and atrial fibrillation | |||
|---|---|---|---|
| Term | Definition | Most common combination | Most common use |
| SAPT (Single Antiplatelet Therapy) |
1 antiplatelet drug | Aspirin | Prevention in CCS in patients without AF |
| DAPT (Dual Antiplatelet Therapy) |
2 antiplatelet drugs | Aspirin + clopidogrel | 6 months after PCI with stent in patients without AF |
| DAT (Dual Antithrombotic Therapy) |
Anticoagulation therapy + antiplatelet therapy | NOAC + clopidogrel | 12 months after PCI in patients with AF |
| TAT (Triple Antithrombotic Therapy) |
Anticoagulation therapy + DAPT | NOAC + Aspirin + clopidogrel |
First week after PCI in patients with AF (on NOAC) |
NOAC – Non-vitamin K Oral Anticoagulant, P2Y12 – P2Y12 adenosine diphosphate (ADP) receptor inhibitor (e.g. Clopidogrel, Prasugrel, Ticagrelor), CCS – Chronic coronary syndrome, PCI – Percutaneous coronary intervention (coronary angioplasty with stent)
ACS after PCI requires antiplatelet therapy; therefore, patients with AF (if they require anticoagulation therapy) and ACS after PCI require:
In ACS and AF, NOAC (not warfarin) are preferred as anticoagulation therapy.
The preferred P2Y12 inhibitor is clopidogrel (not ticagrelor and prasugrel).
Combination of anticoagulation therapy (warfarin or NOAC) with (ticagrelor or prasugrel) is not recommended,
During DAT (warfarin + antiplatelet therapy)
Preferred DAT in AF after ACS or after PCI is:
During TAT and DAT, proton pump inhibitors (pantoprazole) are recommended for prevention of gastrointestinal bleeding.
In patients with AF and stable CCS, anticoagulation therapy alone is recommended (not DAT).
| Anticoagulation and antiplatelet therapy in atrial fibrillation | Class |
|---|---|
| In combination therapy (OAC + antiplatelet therapy), NOAC (not warfarin) are preferred as OAC due to lower bleeding risk and better thromboembolism prevention. | I |
| If the patient is taking (rivaroxaban + antiplatelet therapy), rivaroxaban dose reduction 20 mg → 15 mg once daily may be considered to reduce bleeding risk. | IIa |
| If the patient is taking (dabigatran + antiplatelet therapy), dabigatran dose reduction 150 mg → 110 mg twice daily may be considered to reduce bleeding risk. | IIa |
| If the patient is taking (warfarin + antiplatelet therapy), a target INR 2–2.5 may be considered to reduce bleeding risk. | IIa |
OAC - Oral anticoagulation, NOAC – Non-vitamin K Oral Anticoagulant (Dabigatran, Rivaroxaban, Apixaban, Edoxaban)
| Acute coronary syndrome and atrial fibrillation | Class |
|---|---|
In patients with AF and ACS after PCI (low ischaemic risk), the following is recommended:
|
I |
In patients with AF and ACS after PCI (high ischaemic risk), the following is recommended:
|
IIa |
NOAC – Non-vitamin K oral anticoagulants (Dabigatran, Rivaroxaban, Apixaban, Edoxaban), ACS – Acute coronary syndrome, PCI – Percutaneous coronary intervention
| High ischaemic risk after percutaneous coronary intervention (PCI) | |
|---|---|
| History of stent thrombosis (despite adequate antiplatelet therapy) | |
| Stent implantation in the last remaining patent coronary artery | |
| Diffuse coronary artery disease (especially in patients with diabetes mellitus) | |
| Chronic kidney disease creatinine ≥133 µmol/L (CrCl <60 ml/min) | |
| Implantation of ≥3 stents | |
| Treatment of ≥3 coronary lesions | |
| Treatment of a bifurcation with 2 stents | |
| Total stent length >60 mm | |
| Treatment of CTO (Chronic Total Occlusion) | |
| Chronic coronary syndrome and atrial fibrillation | Class |
|---|---|
In patients with AF and CCS after PCI (low ischaemic risk), the following is recommended:
|
I |
In patients with AF and CCS after PCI (high ischaemic risk), the following is recommended:
|
IIa |
NOAC – Non-vitamin K oral anticoagulants (Dabigatran, Rivaroxaban, Apixaban, Edoxaban), CCS - Chronic coronary syndrome, PCI – Percutaneous coronary intervention
| Duration of antiplatelet therapy after ACS with atrial fibrillation | Class |
|---|---|
| In stable patients after ACS with AF, antiplatelet therapy is not recommended beyond 12 months. | III |
ACS – Acute coronary syndrome
| Antithrombotic therapy in ACS, CCS, and atrial fibrillation | Class |
|---|---|
| NOAC (not warfarin) are the preferred anticoagulation therapy in combination with antiplatelet therapy. | I |
| NOAC doses are reduced according to standard NOAC dose-reduction criteria. | I |
| During treatment with (warfarin + antiplatelet therapy), a target INR 2–2.5 may be considered. | IIa |
| During warfarin therapy (without antiplatelet therapy), a target INR 2–3 is recommended. | I |
| The preferred P2Y12 inhibitor in combination with anticoagulation therapy is clopidogrel (not ticagrelor or prasugrel). | IIa |
ACS – Acute coronary syndrome, CCS - Chronic coronary syndrome, NOAC – Non-vitamin K oral anticoagulants (Dabigatran, Rivaroxaban, Apixaban, Edoxaban)
All of the following patients:

ACS after PCI (Low ischaemic risk)
Example: A patient with AF (on NOAC) had an infarction (STEMI or NSTEMI), was transferred to a cardiac centre where PCI was performed with implantation of 1 stent in the circumflex artery.

ACS after PCI (High ischaemic risk)
Example: A patient with AF had an infarction (STEMI or NSTEMI), was transferred to a cardiac centre where PCI was performed with implantation of 3 stents.

CCS after PCI (Low ischaemic risk)
Example: A patient with AF (on NOAC) had stable angina pectoris and significant coronary stenosis (>70 %) on CT coronary angiography; PCI was performed with implantation of 1 stent in the right coronary artery.

CCS after PCI (High ischaemic risk)
Example: A patient with AF (on NOAC) had stable angina pectoris; coronary angiography showed 3 severe stenoses (>70 %), and 3 stents were implanted during PCI.

Stable CCS
Example: A patient with AF (on NOAC) has CCS with non-significant coronary stenosis (<50 %), not indicated for PCI.
In patients after PCI, bleeding risk is assessed using the ARC-HBR (Academic Research Consortium – High Bleeding Risk) score.
| ARC-HBR score (Bleeding risk) |
|---|
Major criteria (1 is sufficient)
|
Minor criteria (≥2 required)
|
ARC-HBR - Academic Research Consortium – High Bleeding Risk. PCI - Percutaneous coronary intervention. NOAC – Non-vitamin K Oral Anticoagulant (Dabigatran, Rivaroxaban, Apixaban, Edoxaban). eGFR = estimated Glomerular Filtration Rate. NSAIDs - non-steroidal anti-inflammatory drugs
| Bleeding risk according to ARC-HBR score and shortening of antithrombotic therapy after PCI | ||
|---|---|---|
| ARC-HBR score | Major bleeding risk (within 1 year after PCI) |
Antithrombotic therapy after PCI |
| Positive (≥ 1 major criterion or ≥ 2 minor criteria) |
4 – 9 % |
|
| Negative | 1 – 3 % |
|
ARC-HBR - Academic Research Consortium – High Bleeding Risk. PCI - Percutaneous coronary intervention. DAT - Dual Antithrombotic Therapy. TAT - Triple Antithrombotic Therapy.
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
