

Classification:
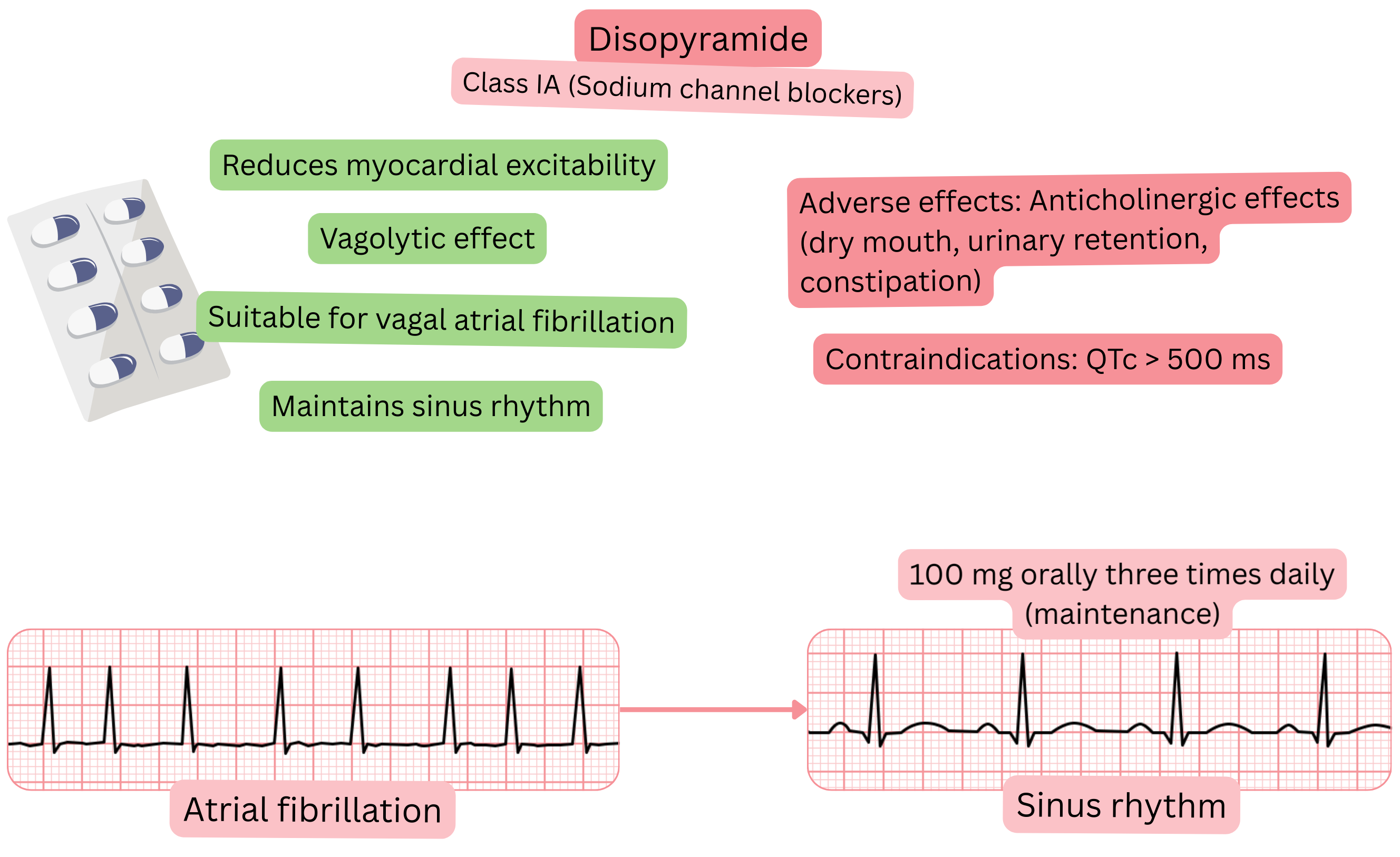
Mechanism:
Effect on AF:
| Disopyramide and atrial fibrillation (AF) |
|---|
| Brand names |
| Disopyramide, Disopiramida, Disopyramidum, Dirytmin, Durbis, Rythmodan |
| Indications |
|
| Dosing |
|
| Onset of action |
|
| Effect |
Maintenance of sinus rhythm (paroxysmal or persistent AF) at 1 year
|
| Duration of action |
|
| Contraindications |
|
Patient monitoring during disopyramide therapy:
| Patient monitoring after initiation of disopyramide | ||
|---|---|---|
| Time from initiation | What to monitor | Reason for treatment discontinuation |
| Week 1 |
ECG (QTc, QRS) Renal function, electrolytes (K⁺, Mg²⁺) Anticholinergic symptoms |
QTc > 500 ms QRS widening > 25% Anticholinergic symptoms Arrhythmias (torsades de pointes) Electrolyte or renal impairment |
| 3 months |
ECG (QTc, QRS) Renal function, electrolytes (K⁺, Mg²⁺) Anticholinergic symptoms |
QTc > 500 ms QRS widening > 25% Anticholinergic symptoms Arrhythmias (torsades de pointes) Electrolyte or renal impairment |
| 6–12 months |
Echocardiography Long-term treatment tolerance |
Ejection fraction < 40 % Poor tolerance or lack of treatment efficacy |
Adverse effects:
Disopyramide and procainamide belong to Class IA anti-arrhythmic drugs but have different properties.
| Disopyramide vs Procainamide and atrial fibrillation | ||
|---|---|---|
| Property | Disopyramide | Procainamide |
| Class | IA + anticholinergic (vagolytic) effect | IA (Na⁺ channel blocker) |
| Route of administration | Oral (chronic prevention of recurrences) | Intravenous (acute conversion of AF, especially with WPW) |
| Pre-excited AF (WPW) | No – not suitable | Yes – effective and safe |
| Vagal AF | Yes – first-line drug, suppresses vagal tone | Neutral effect, no vagolytic activity |
| Maintenance of sinus rhythm | Effective mainly in vagal AF | No – not used for long-term therapy |
| Main risks |
QT prolongation, torsades de pointes, anticholinergic adverse effects (dry mouth, urinary retention) |
Hypotension, QT prolongation, lupus-like syndrome |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
