

Classification:

Mechanism:
Effect on AF:
| Diltiazem and atrial fibrillation (AF) |
|---|
| Brand names |
| Diacordin, Cardizem, Tildiem, Dilzem, Altiazem, Adizem, Angitil |
| Indications |
|
| Dosing |
|
| Onset of action |
|
| Effect |
|
| Duration of action |
|
| Contraindications |
|
Patient monitoring after initiation of diltiazem:
| Patient monitoring after initiation of diltiazem | ||
|---|---|---|
| Time since initiation | What to monitor | Reason for treatment interruption |
| Week 1 |
ECG Blood pressure |
AV block II or III degree Bradycardia < 50/min. Hypotension < 100/60 mmHg |
| 6–12 months | Echocardiography | Ejection fraction < 40 % |
Adverse effects
Verapamil and diltiazem belong to Class IV anti-arrhythmic drugs, but they are different molecules,
| Verapamil vs. diltiazem in atrial fibrillation (AF) | ||
|---|---|---|
| Characteristic | Verapamil | Diltiazem |
| Effect on AV node | Stronger (slows AF more) | Weaker (slows AF less) |
| Negative inotropy | More pronounced (caution if EF<50%) | Milder (better tolerability) |
| Peripheral vasodilation | Weaker (lower risk of hypotension) | Stronger (higher risk of hypotension) |
| Typical adverse effects | Constipation, bradycardia, AV block | Ankle oedema, hypotension |
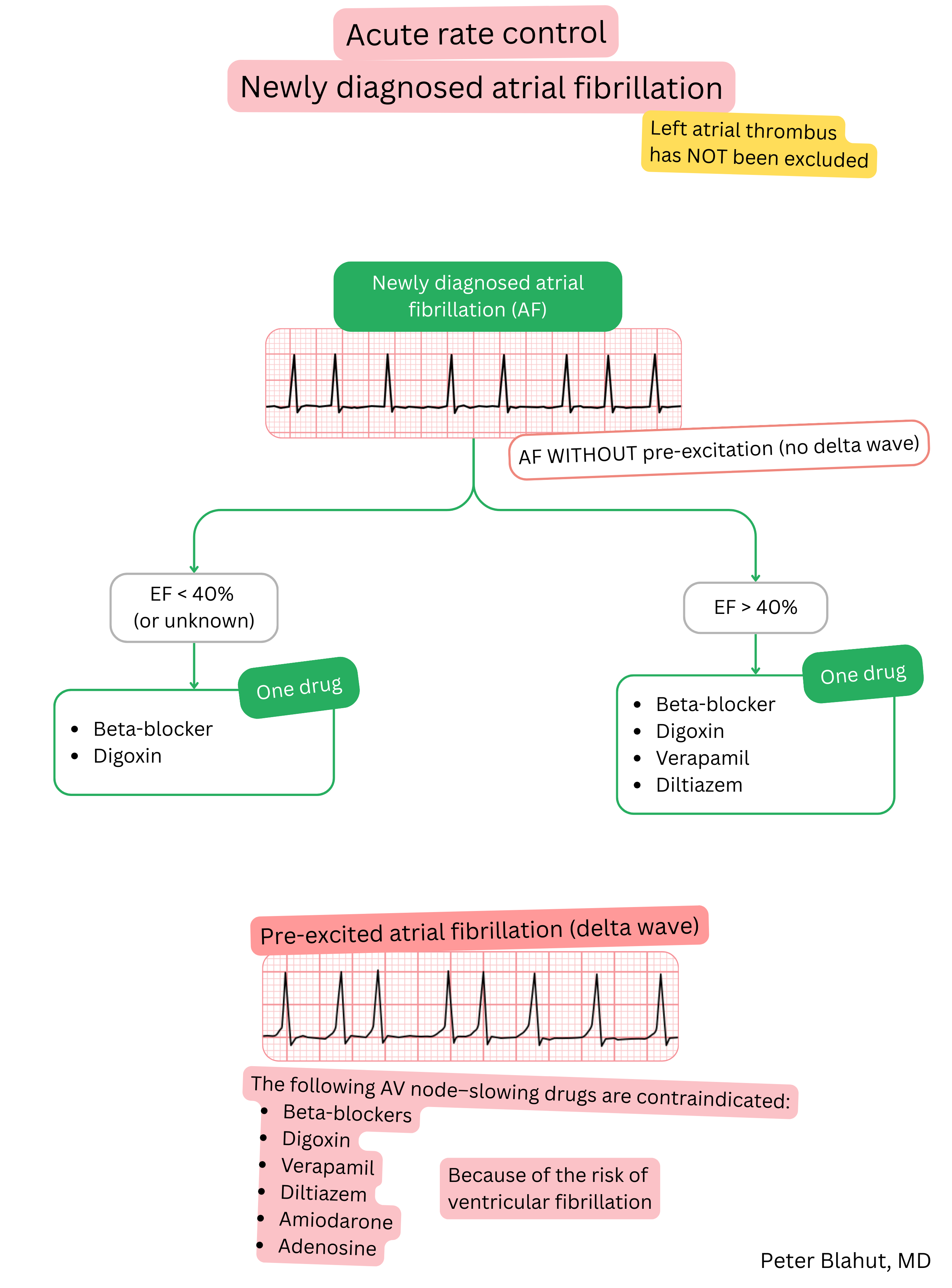
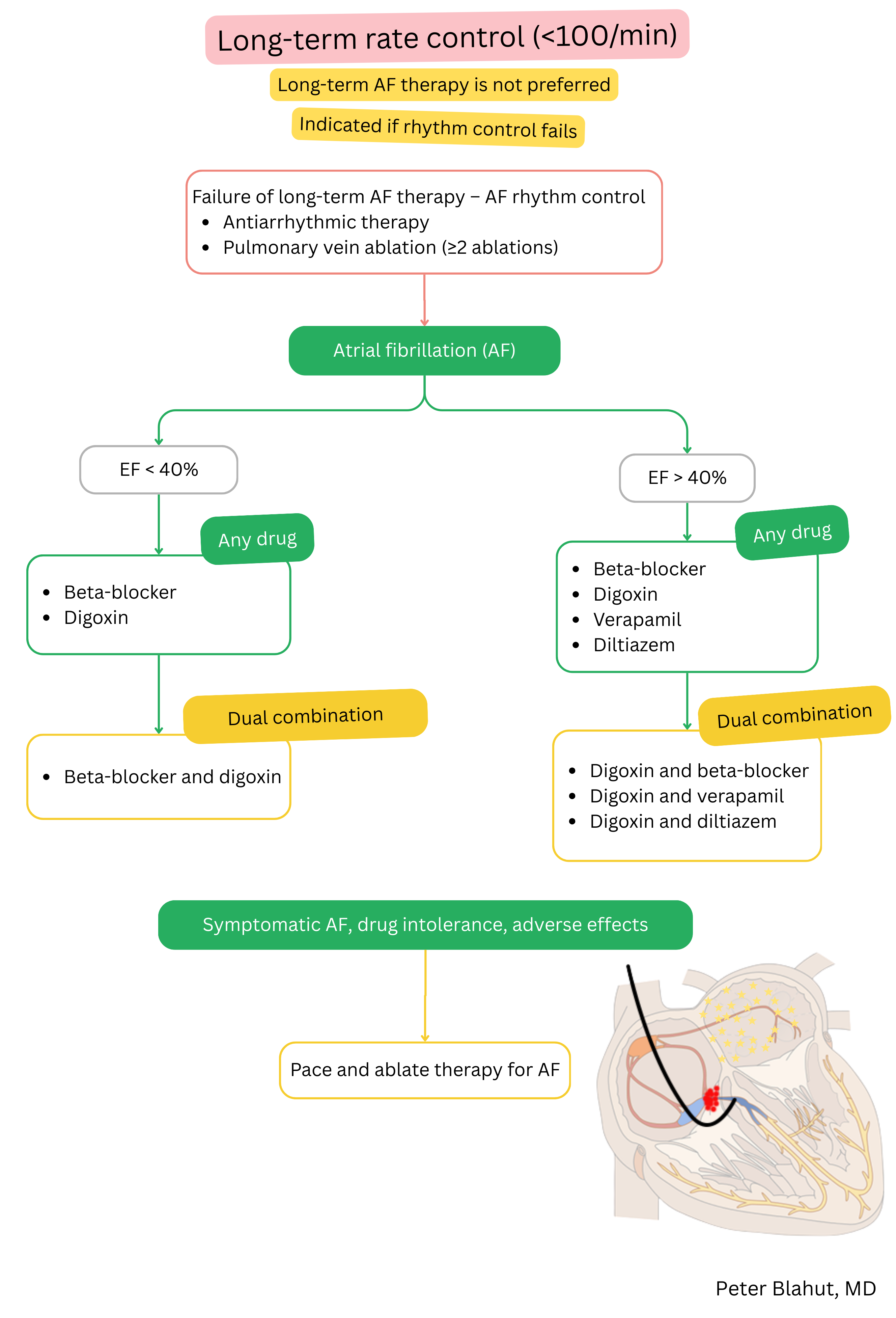
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
