

Classification:
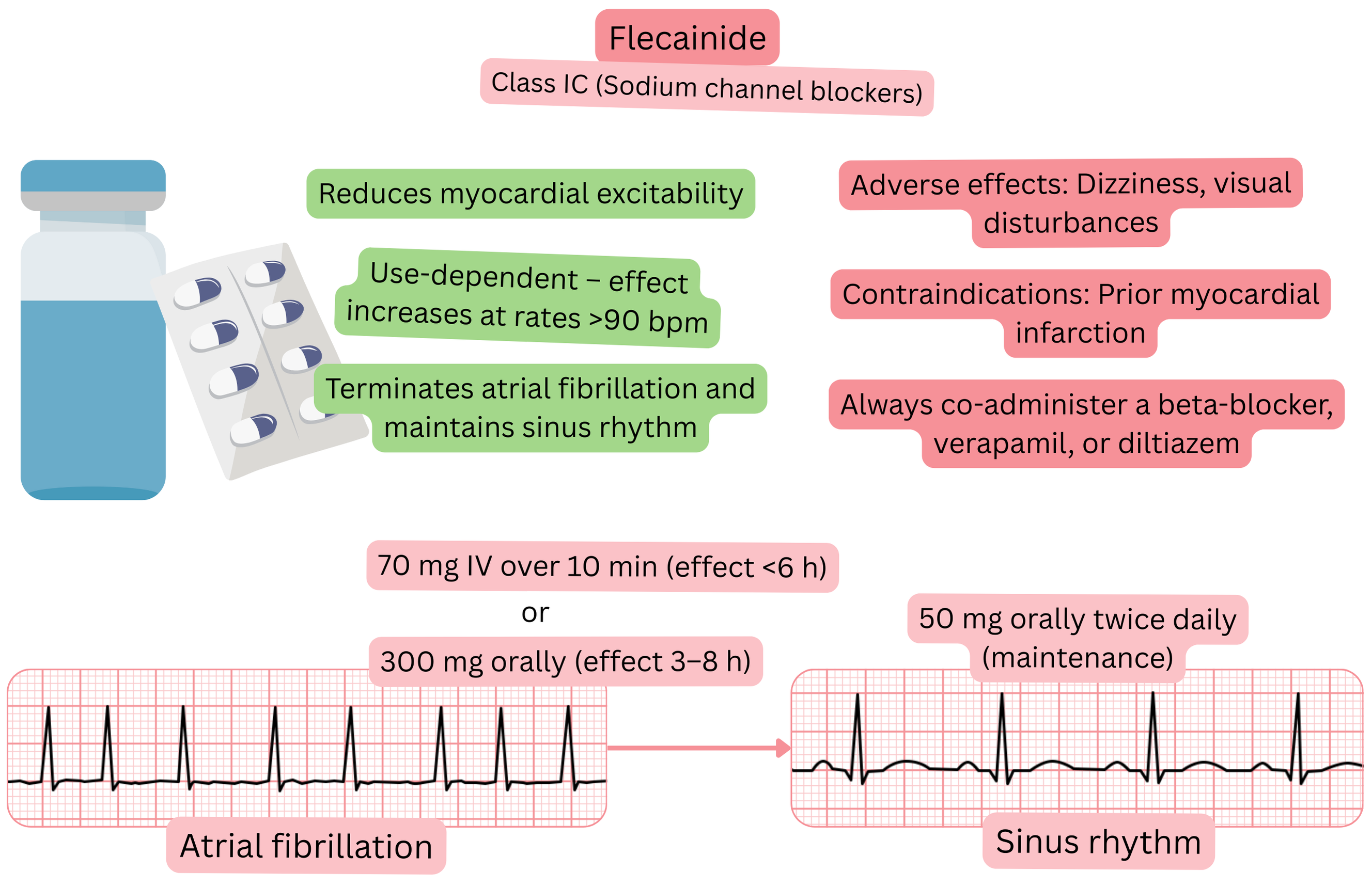
Mechanism:
Effect on AF:
| Flecainide and atrial fibrillation (AF) |
|---|
| Brand names |
| Amarhyton, Flekainid, Tambocor, Apocard, Almarytm, Flécaïne, Flecaine, Flecadura, Juneflecad |
| Indications |
|
| Dosing |
|
| Onset of action |
|
| Effect |
Time to conversion to sinus rhythm and success rate
|
| Duration of action |
|
| Contraindications |
|
Patient monitoring after initiation of flecainide:
| Patient monitoring after initiation of flecainide | ||
|---|---|---|
| Time since initiation | What to monitor | Reason for treatment discontinuation |
| 1 week |
ECG (QRS, PR interval) Blood pressure |
QRS > 120 ms or prolongation > 25 % Bradycardia < 50/min Hypotension < 90/60 mmHg Brugada pattern on ECG |
| 1 month | ECG (QRS, PR interval) |
QRS > 120 ms or prolongation > 25 % Brugada pattern on ECG |
| 6–12 months |
ECG (QRS, PR interval) Laboratory tests Echocardiography Holter ECG as needed |
Ejection fraction < 40 % QRS > 120 ms or prolongation > 25 % Brugada pattern on ECG Severe laboratory abnormalities |
Adverse effects:
Propafenone and flecainide both belong to Class IC anti-arrhythmic drugs, but they are distinct molecules.
| Propafenone vs. flecainide and atrial fibrillation (AF) | ||
|---|---|---|
| Property | Propafenone | Flecainide |
| Mechanism of action | Na+ channel blockade + mild beta-blocking effect | Na+ channel blockade, without beta-blocking effect |
| Effect on AV node | Mild slowing of conduction (via beta-blockade) | Virtually no direct effect |
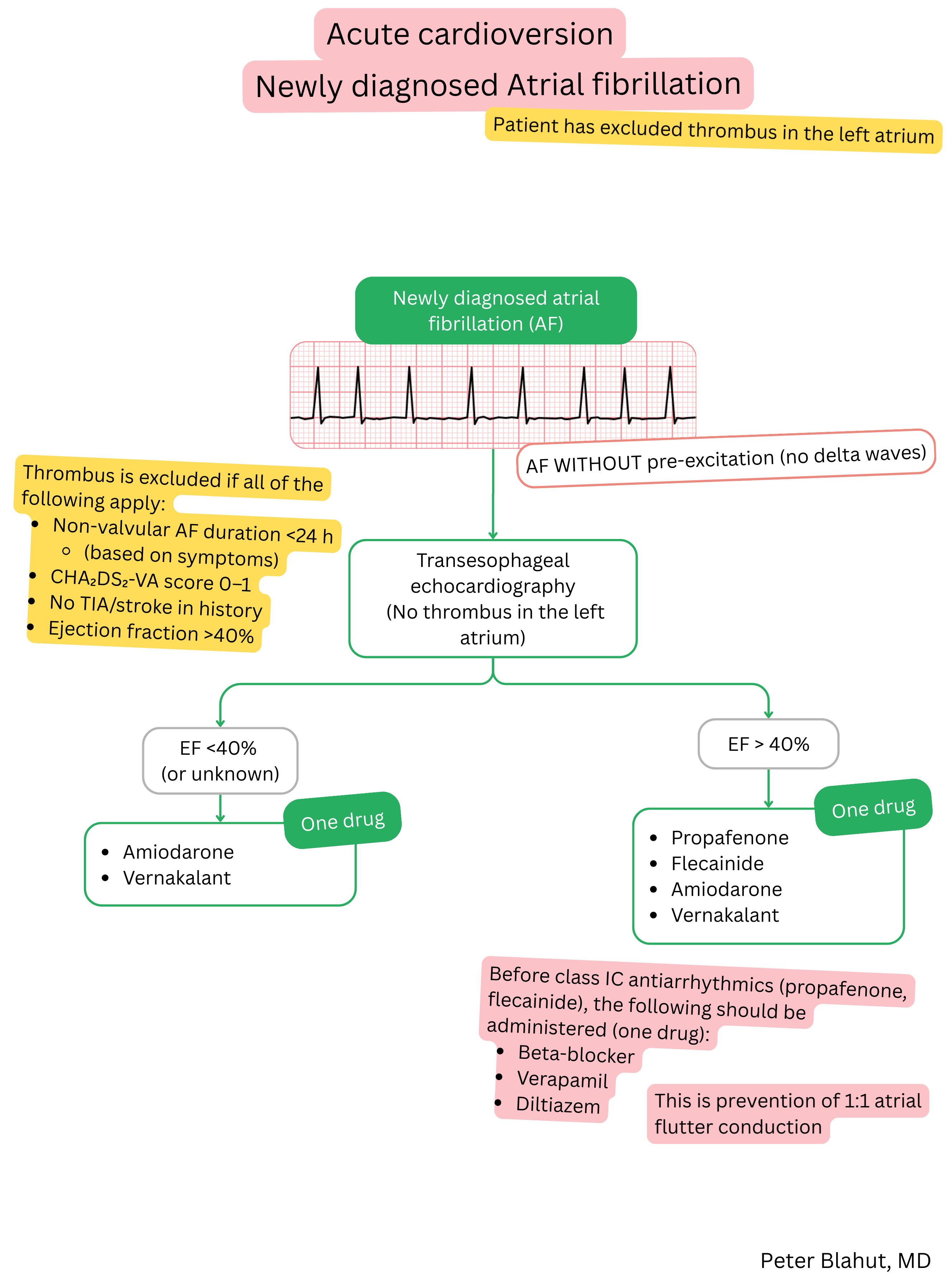
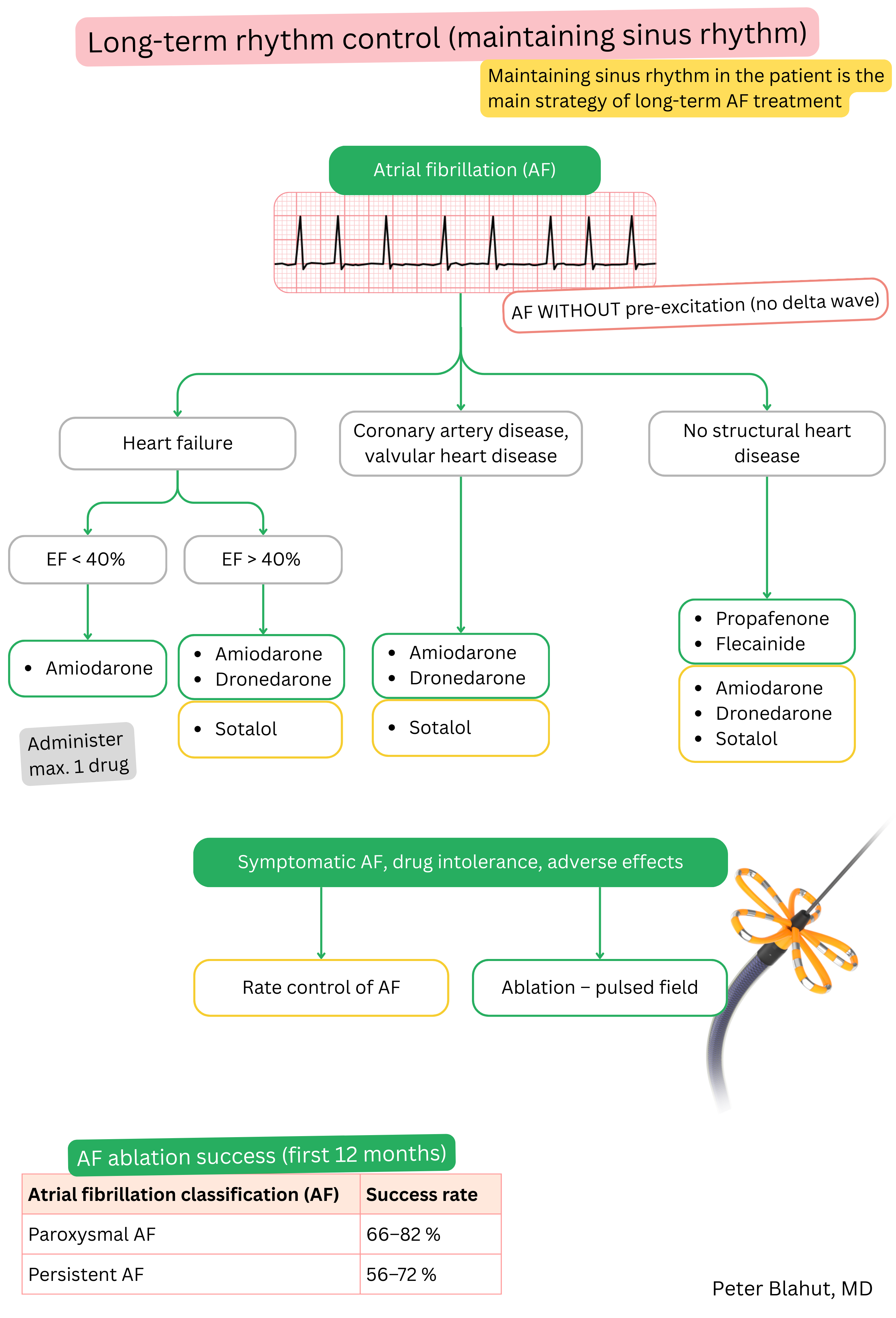
| Use in AF | Acute cardioversion, maintenance of sinus rhythm, partial rate control | Acute cardioversion, maintenance of sinus rhythm (combination with BB/NDHP CCB is recommended) |
| Efficacy at 1 year | ~40–60 % maintenance of sinus rhythm | ~50–65 % maintenance of sinus rhythm |
| Effect on heart rate | Reduces (due to beta-blockade) | No significant effect |
| Use-dependence | Less pronounced; partially attenuated by beta-blocking effect | Pronounced; marked QRS prolongation at higher heart rates |
| Adverse effects | Nausea, metallic taste, bradycardia, hypotension | Dizziness, visual disturbances |
These guidelines are unofficial and do not represent formal guidelines issued by any professional cardiology society. They are intended for educational and informational purposes only.
