The following concepts and principles are key for antiarrhythmic pharmacological treatment of atrial fibrillation (AF):
- Action potential
- Nodal action potential
- Non-nodal action potential
- Use-dependent antiarrhythmic drugs (Class IC – Propafenone, Flecainide)
- Reverse use-dependent antiarrhythmic drugs (Class III – Sotalol – strongest, Amiodarone, Dronedarone)
- Effective refractory period (Class III and IA)
- Autonomic nervous system
- Sympathetic nervous system (Class II – beta-blockers)
- Parasympathetic nervous system (Digoxin)
Action potential
- Each cardiomyocyte in the heart has an electrical voltage difference between the outer and inner sides of the membrane.
- This voltage arises due to different ion concentrations (mainly Na⁺, K⁺, and Ca²⁺) on both sides of the membrane
- and its selective permeability maintained by the Na⁺/K⁺-ATPase.
- During the cardiac cycle, ions move across the membrane and the electrical voltage changes accordingly.
- The change in electrical voltage during the cardiac cycle is represented by the action potential (AP) curve.
- The AP arises spontaneously in the SA node and subsequently spreads through adjacent cardiomyocytes to the atria.
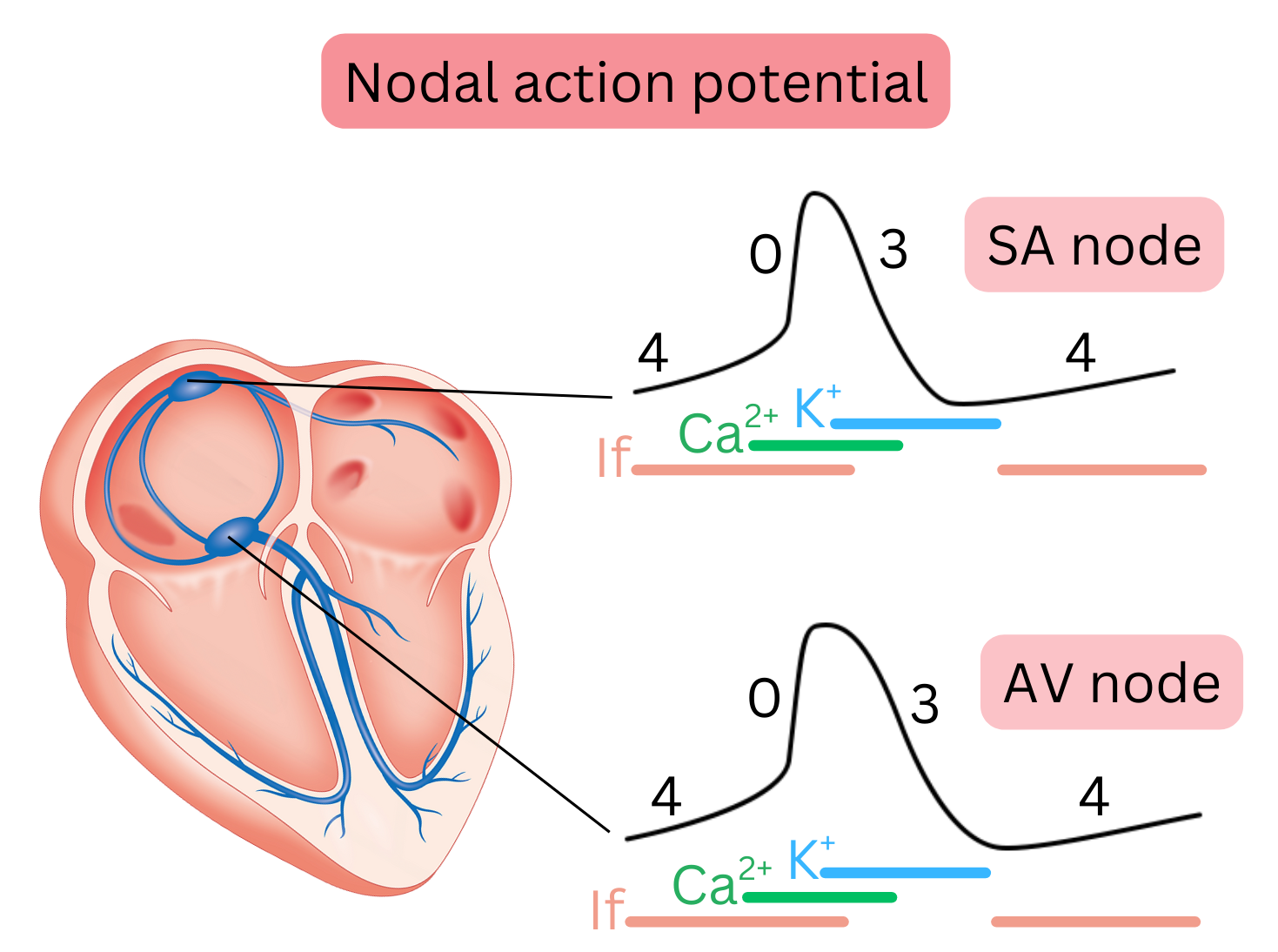
Nodal action potential
- Also referred to as pacemaker action potential (AP).
- It is present in the SA node and AV node and is therefore termed nodal AP.
- It arises spontaneously, i.e. spontaneous depolarization occurs repeatedly.
- Spontaneous depolarization is mediated by If currents, during which Na⁺ ions cross the membrane.
- It is a slow AP because Ca²⁺ ions cross the membrane slowly during depolarization.
- It has a slow onset (depolarization) but short duration, so a new AP is generated relatively quickly.
- It has 3 phases (4, 0, 3), during which specific ions cross the membrane in each phase.
| Nodal action potential and antiarrhythmic drugs |
| Drug |
Class |
Mechanism |
SA node |
AV node |
| β-blockers |
II |
↓ sympathetic tone |
↓ rate |
↓ conduction |
| Ca-blockers |
IV |
Ca²⁺ blockade |
↓ rate |
↓ conduction |
| Digoxin |
– |
↑ parasympathetic tone |
↓ rate |
↓ conduction |
| Ivabradine |
– |
If blockade |
↓ rate |
– |
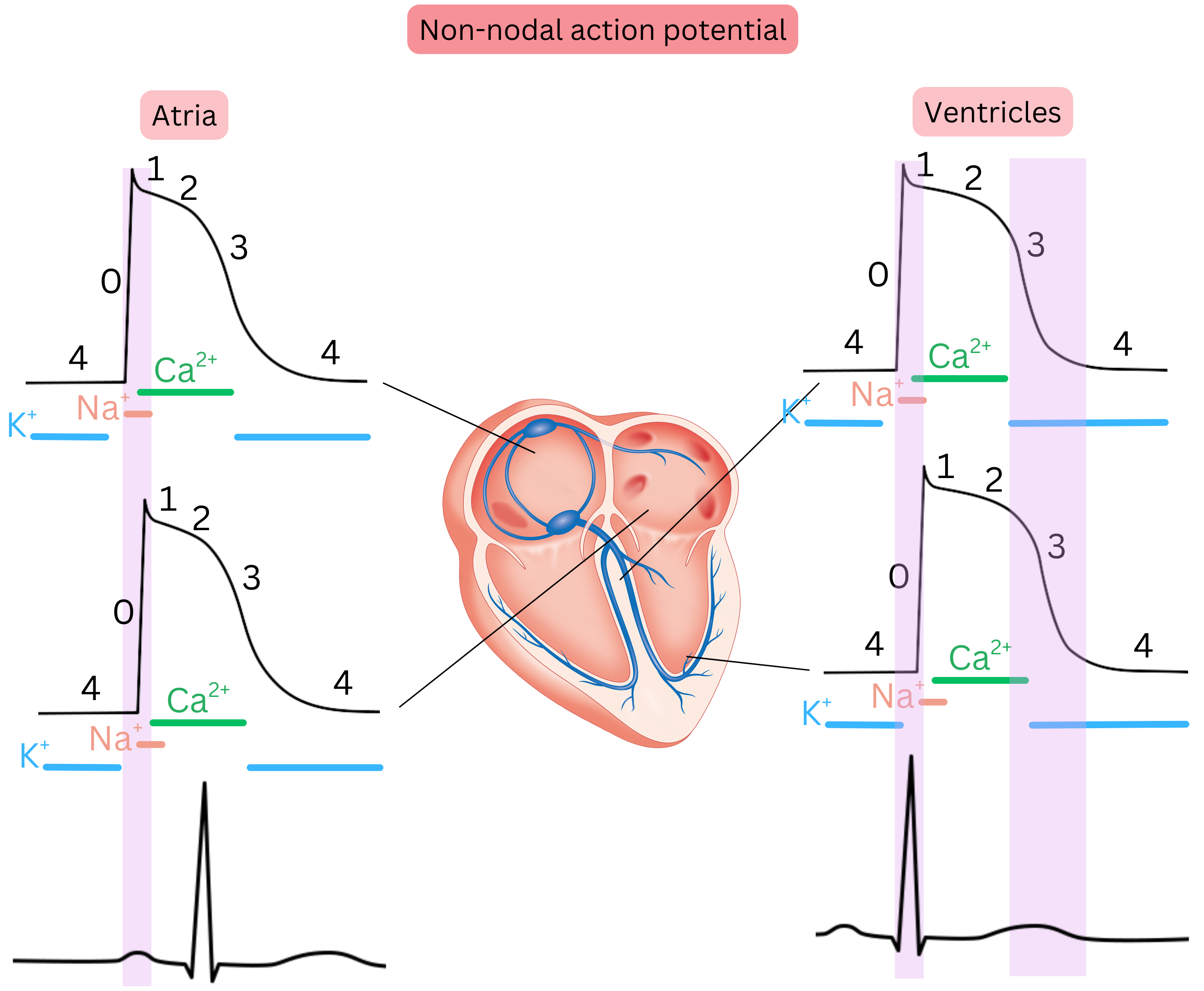
Non-nodal action potential
- Also referred to as non-pacemaker action potential (AP).
- It is present in the working myocardium of the atria, ventricles, and in Purkinje fibres.
- It does not depolarize spontaneously; it requires an AP from an adjacent cardiomyocyte to trigger it.
- It is fast because Na⁺ ions cross the membrane rapidly during depolarization.
- It has 5 phases (0, 1, 2, 3, 4), during which specific ions cross the membrane in each phase.
| Non-nodal action potential (AP) and antiarrhythmic drugs |
| Drug |
Class |
Mechanism |
AP |
ECG (QRS/QT) |
| Quinidine, Procainamide, Disopyramide |
I A |
Na⁺ + K⁺ channel blockade |
↑ AP |
↑ QT |
| Lidocaine, Mexiletine |
I B |
Na⁺ channel blockade (ischaemic tissue) |
↓ AP |
↓ QT |
| Flecainide, Propafenone |
I C |
Strong Na⁺ channel blockade |
≈ AP |
↑ QRS, QT ≈ |
| Amiodarone, Sotalol, Dronedarone |
III |
K⁺ channel blockade |
↑ AP |
↑ QT |
The non-nodal AP spreads progressively from the SA node across the atrial myocardium, and atrial depolarization (phase 0) is seen on the ECG as the P wave. The depolarization wave crosses the atria in < 100 ms; therefore, the P wave duration is < 100 ms.
The non-nodal AP spreads from the atria through the AV node to the Purkinje fibres and ventricles. Ventricular depolarization (phase 0) is seen on the ECG as the QRS complex. The depolarization wave crosses the ventricles in < 110 ms; therefore, QRS duration is < 110 ms.
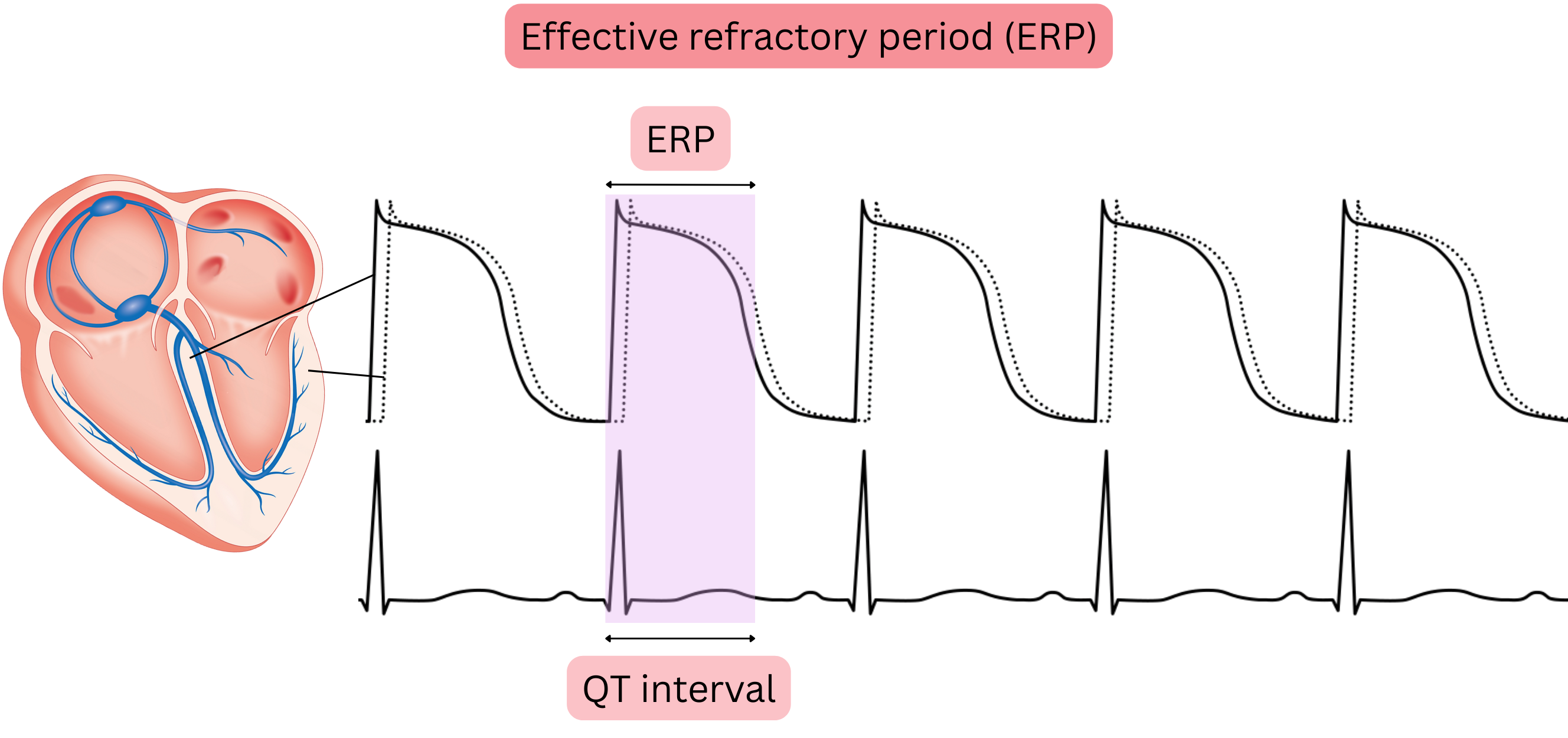
Effective refractory period (ERP)
- The time from the onset of depolarization (phase 0) to almost the end of repolarization (phase 3)
- During the ERP, no further depolarization, i.e. no additional action potential, can occur in cardiomyocytes,
- because Na⁺ channels must return to the resting state after depolarization before they can be activated again.
- The duration of the ERP is reflected on the ECG as the QT interval.
- The QT interval represents the duration of the non-nodal AP
- ERP (QT interval) is prolonged mainly by Class IA and Class III antiarrhythmic drugs,
- because they block K⁺ channels and thereby slow repolarization.
- A longer ERP means that the myocardium remains non-excitable for a longer time, which prevents rapid re-propagation of impulses
- thereby reducing the maximum ventricular rate during tachy-AF and
- preventing re-entry.
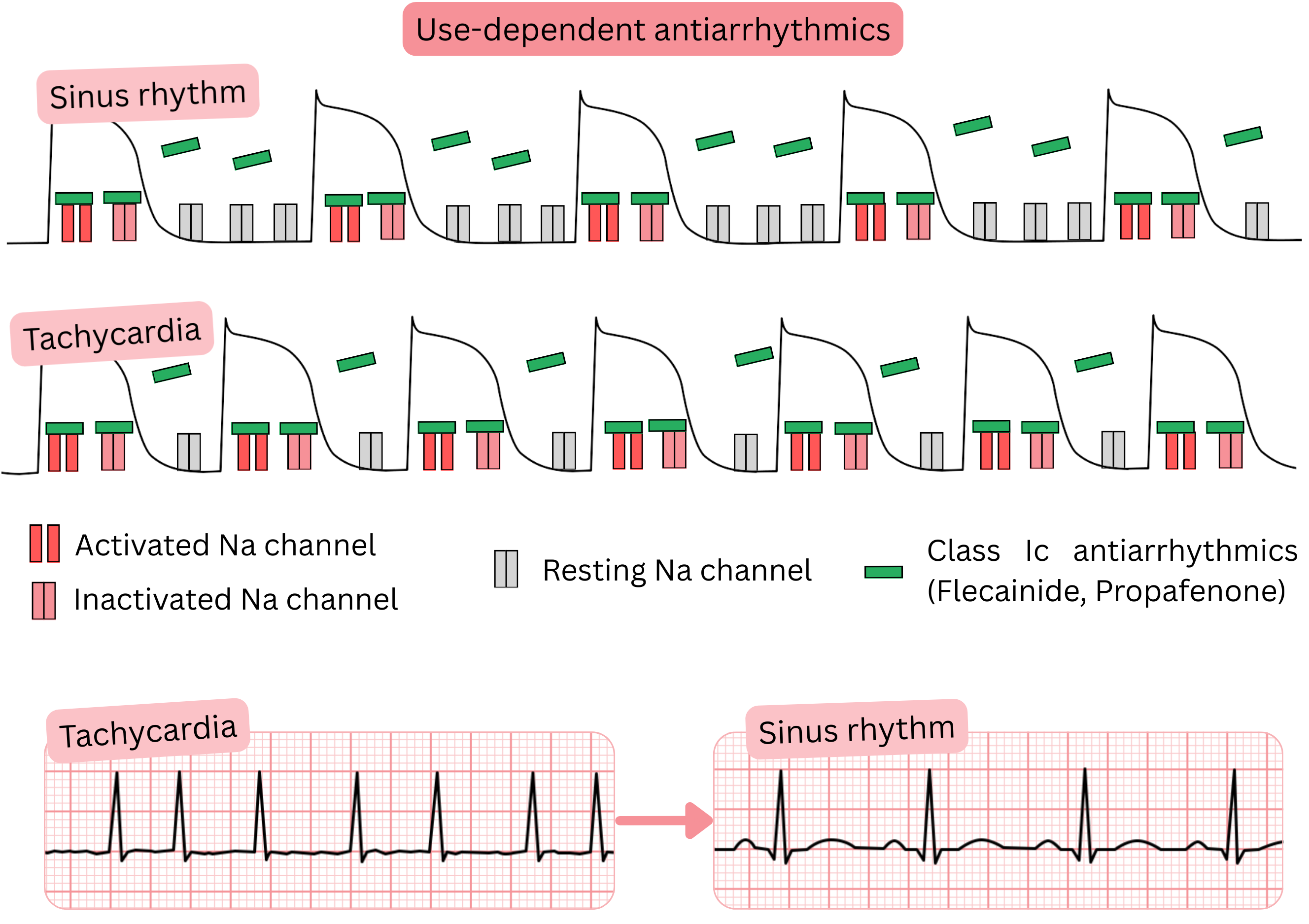
Use-dependent antiarrhythmic drugs
- Use dependence refers to antiarrhythmic drugs that bind to ion channels
- more intensively at higher heart rates (> 90/min)
- This includes Class IC antiarrhythmic drugs (Propafenone, Flecainide), which in AF are used for:
- pharmacological cardioversion of tachy-AF (> 100/min) to sinus rhythm
- maintenance of sinus rhythm (rhythm control)
- Mechanism of action (use-dependent antiarrhythmic drugs):
- They bind preferentially to activated and inactivated Na⁺ channels
- Higher heart rate (> 90/min) → greater degree of blockade (use dependence)
- During tachycardia, diastole (phase 4 of the action potential) shortens,
- so Na⁺ channels remain longer in the activated or inactivated state
- Class IC antiarrhythmic drugs therefore remain bound to Na⁺ channels for longer → higher cumulative effect.
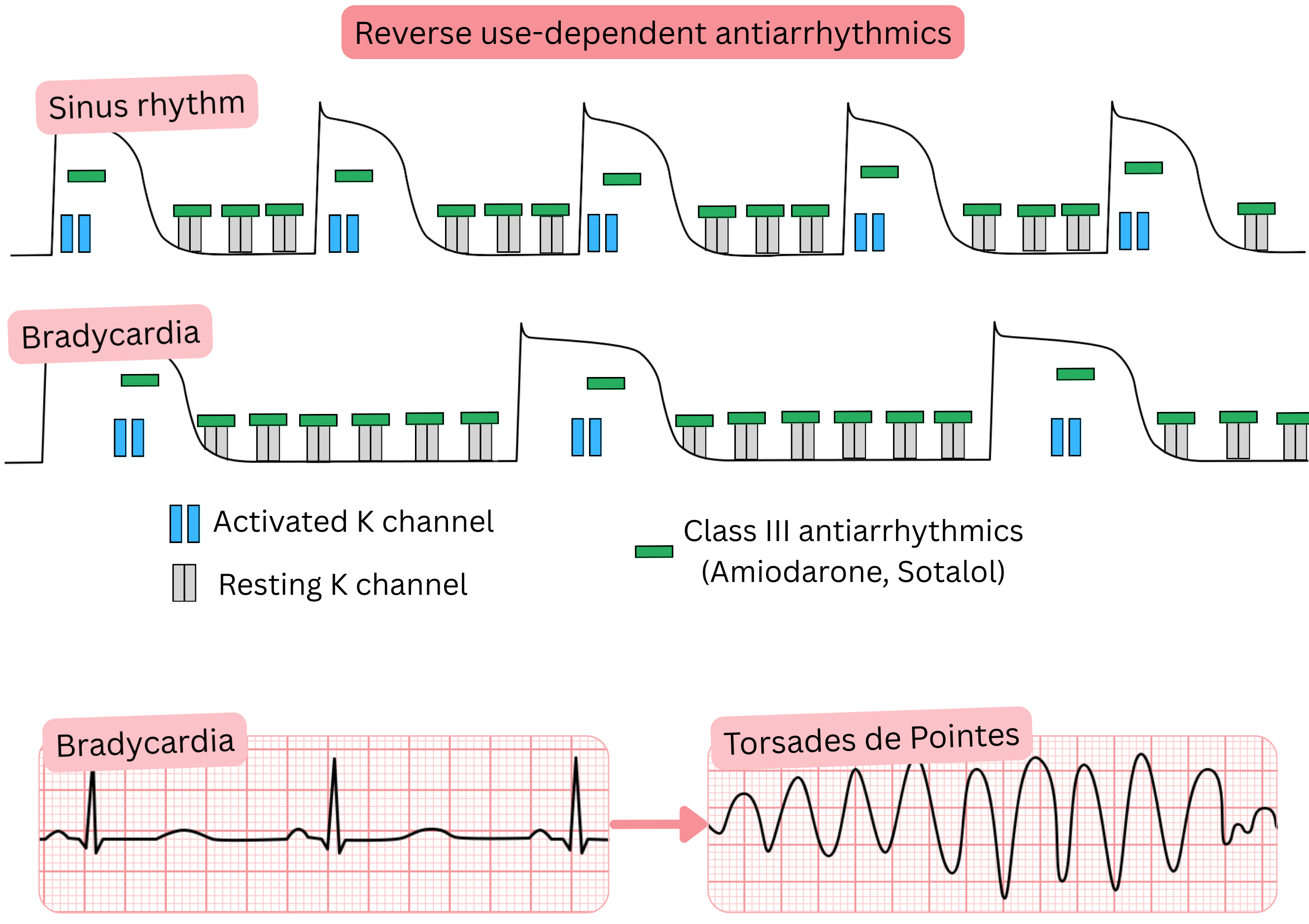
Reverse use-dependent antiarrhythmic drugs
- Reverse use dependence refers to antiarrhythmic drugs that bind to ion channels
- more intensively at lower heart rates (< 90/min)
- This includes Class III antiarrhythmic drugs (Sotalol – strongest, Amiodarone, Dronedarone)
- In AF, they are used for:
- maintenance of sinus rhythm (rhythm control)
- Mechanism of action (reverse use-dependent antiarrhythmic drugs):
- They bind preferentially to K⁺ channels (phase 4) and block K⁺ channels
- Subsequently, K⁺ channels are blocked also in phase 3, resulting in QT interval prolongation
- Lower heart rate (bradycardia) → greater degree of blockade (reverse use dependence)
- At slower heart rates, diastole (phase 4) and the entire action potential (phase 3) are prolonged
| Use dependence and reverse use dependence antiarrhythmic drugs |
| Class |
Antiarrhythmic drugs |
Mechanism |
Type |
ECG effect |
When to discontinue |
| I A |
Quinidine,
procainamide,
disopyramide |
Na⁺ and K⁺ channel blockade |
Use dependence |
↑ QT;
↑ QRS/PR (± mild) |
QTc > 500 ms or
ΔQTc > 60 ms;
QRS ↑ ≥ 25 % or
> 120 – 130 ms |
| I B |
Lidocaine,
mexiletine |
Na⁺ channel blockade (ischaemic tissue) |
Use dependence |
↓ QT;
QRS ≈;
PR ≈ |
QRS ↑ ≥ 25 % from baseline
or BBB |
| I C |
Flecainide,
propafenone |
Strong Na⁺ channel blockade |
Use dependence |
↑ QRS;
QT ≈;
PR ≈/↑ |
QRS ↑ ≥ 25 % or
> 120 – 130 ms;
PR > 240 ms;
new BBB/AV block |
| III |
Sotalol,
dofetilide,
ibutilide |
K⁺ channel blockade |
Reverse use dependence |
↑ QT (risk of TdP at HR < 50/min) |
QTc > 500 ms or ΔQTc > 60 ms;
HR < 50 – 60/min |
| III |
Amiodarone |
K⁺, Na⁺, Ca²⁺ channel blockade + β-blockade |
Reverse use dependence (mild) |
↑ QT (mild);
± ↑ PR/QRS |
QTc > 500 ms;
HR < 50/min;
AV block, BBB |
| III |
Dronedarone |
K⁺, Na⁺, Ca²⁺ channel blockade + β-blockade (weaker) |
Reverse use dependence (mild) |
↑ QT (mild) |
QTc > 500 ms;
HR < 50/min;
AV block, BBB |
BBB – Bundle Branch Block (RBBB or LBBB), TdP – Torsades de Pointes
The autonomic nervous system has two main opposing components:
- Sympathetic nervous system
- Parasympathetic nervous system
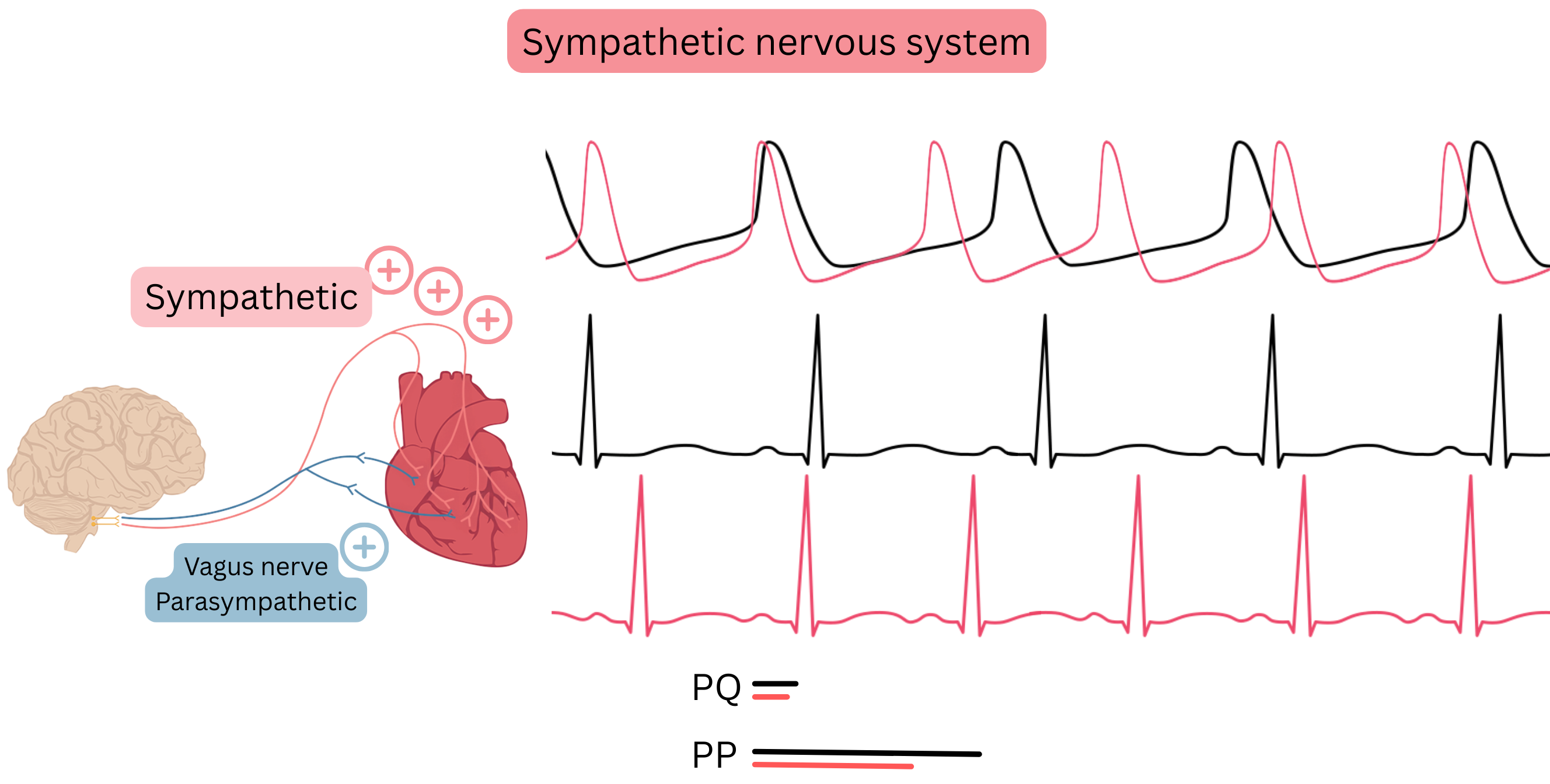
Sympathetic nervous system
- The main mediators of the sympathetic nervous system are catecholamines, which bind to adrenergic receptors
- Catecholamines (adrenaline, noradrenaline, dopamine)
- Adrenergic receptors (α1, α2, β1, β2, β3)
- For antiarrhythmic drugs, beta receptors are particularly relevant:
- β1 – located in the heart, predominantly in the SA node and less in the AV node
- β2 – located in bronchi, lungs, and vessels
- The sympathetic nervous system is mainly targeted by Class II antiarrhythmic drugs (beta-blockers)
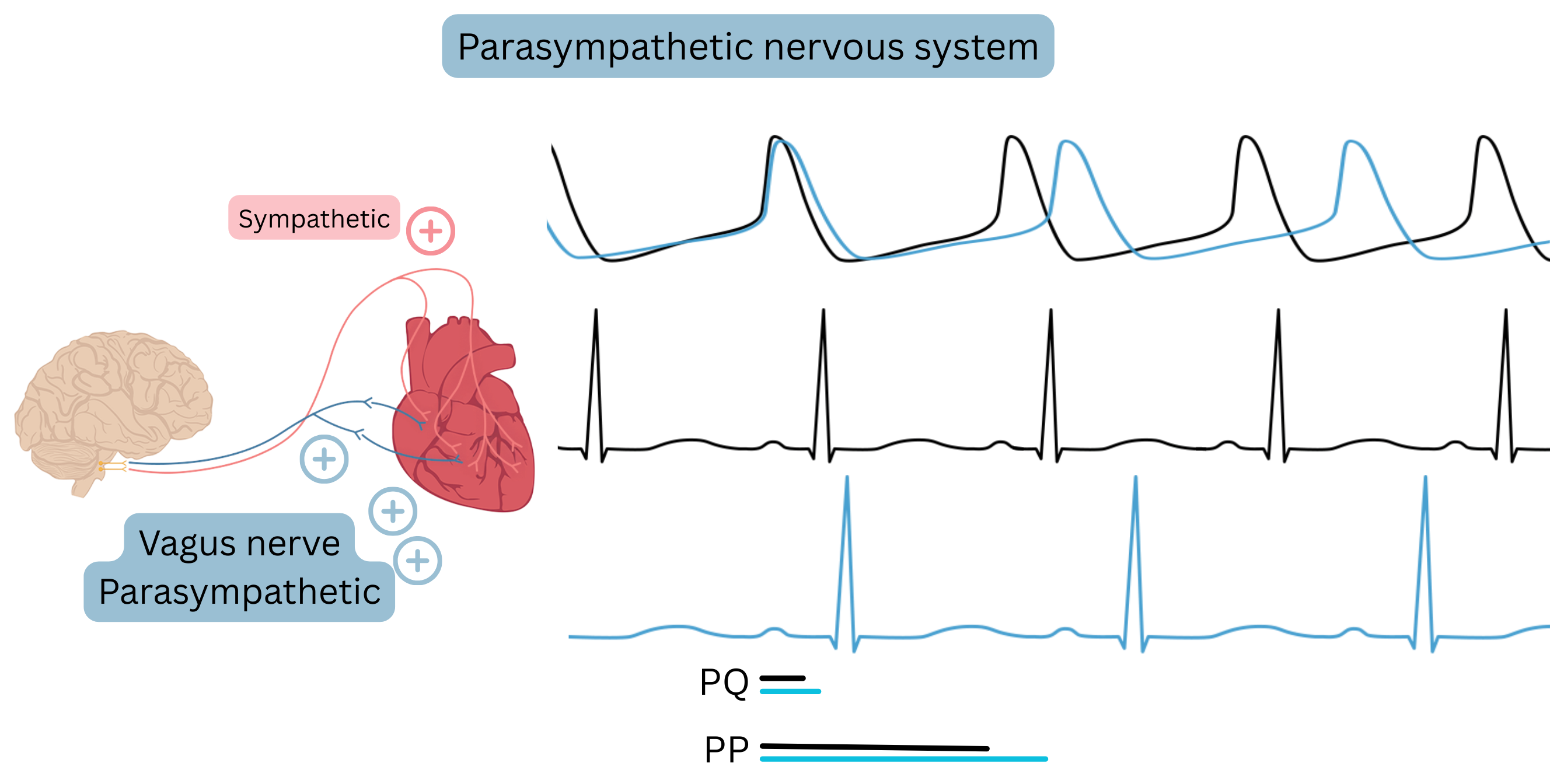
Parasympathetic nervous system
- The main parasympathetic nerve is the vagus nerve
- It innervates the pupils, salivary glands, bronchi, gastrointestinal tract, urinary bladder, and the heart
- In the heart, the vagus predominantly innervates the AV node and less the SA node; it also innervates the atrial myocardium and minimally the ventricles
- The SA node is innervated via the right vagus
- The AV node is innervated via the left vagus
- therefore, for termination of supraventricular tachycardia (AVNRT, AVRT), massage of the left carotid sinus is more effective.
- The parasympathetic nervous system is mainly influenced by digoxin
| Autonomic nervous system and antiarrhythmic drugs |
| Drugs |
Effect on the nervous system |
Mechanism |
Effect |
| β-blockers |
Inhibit sympathetic tone |
β1 (± β2) receptor blockade |
↓ SA rate; ↓ AV conduction |
| Digoxin |
Stimulates parasympathetic tone |
↑ vagal tone |
↓ AV conduction; ± ↓ SA rate |









